
Category Archives: Pharmaceuticals


Myeloid Therapeutics rebrands and unveils an expanded strategy to move beyond its previous myeloid cell focus
- Proof-of-mechanism: Paired biopsies confirmed CAR+ immune cells infiltrating tumors, with immune remodeling and CD8 T cell recruitment.
- Safety and repeat dosing: Over 200 doses delivered with a consistent, manageable safety profile and no cumulative toxicities.
- Evidence of activity: CAR expression detected in circulating immune cells, with stable disease in several patients and a confirmed partial response on treatment for 16 months.
- MT-302 (TROP2; solid tumors): Dose escalation completed; tolerable safety profile.
- MT-303 (GPC3; hepatocellular carcinoma): Dose escalation ongoing.
- MT-304 (HER2; solid tumors): First patient expected Q4 2025; first-in-class multi-immune CAR engaging NK and myeloid cells.
- Retrotransposon-based in vivo CAR-T: First all-RNA product candidate with permanent CAR integration for B-cell depletion.
- Additional multi-lineage programs in preclinical development across oncology and immunology.
- Selective activation: Cell-specific receptors and LNPs for precise T, myeloid, and NK cell programming.
- Flexible durability: Transient or stable CAR expression, with permanent integration via RNA-based retrotransposon technology.
- Industry-leading RNA: Up to 8+ days of linear mRNA expression with no reactogenicity.
- Speed and scalability: Concept-to-clinic in <12 months with low-cost manufacturing.
9th Annual Patients as Partners® Europe Event to Focus on Advancing Patient Involvement in Clinical Research

NEW YORK, NY, United States, 11-Feb-2025 — /EuropaWire/ — The Conference Forum has announced the 9th annual Patients as Partners® Europe event, set for May 20-21, 2025, at the Royal National Hotel in London. This event provides a unique platform for pharma R&D professionals and patient advocates to discuss how patient involvement can reduce risks and inefficiencies in clinical research while advancing the development of new treatments.
Alfred Samuels, a patient advocate and co-chair for 2025, emphasized the importance of recognizing patients as experts in their conditions, which leads to more successful trials and better healthcare outcomes.
Keynote speakers include:
- Harry Verbunt, who will share his post-cancer clinical trial experience and suggestions for improving trial access.
- Peter DiBiaso, discussing the importance of placing patients at the center of research.
- Dr. Temi Olonisakin, offering patient perspectives on support in clinical research.
- Lea-Isabelle Proulx, PhD (Roche), on reducing patient burden through improved protocols.
- Beyza Klein (Johnson & Johnson), on embedding a patient engagement strategy.
- Dr. Catherine Coulouvrat (Sanofi), discussing how patient experience data informs product tolerability.
- Marisa Minetti (Chiesi Farmaceutici), on leveraging patient archetypes in protocol development.
- Gisela Linthorst (Azafaros), on involving the patient community in early clinical trials.
Pharma companies such as AstraZeneca, Boehringer-Ingelheim, GSK, and others will also present case studies on advancing patient partnerships.
The event will explore critical topics including patient-informed research, decentralized trials, increasing trial representation, and reducing patient burden through technology.
Patients as Partners® Europe is co-produced with patients, R&D, academia, and non-profit organizations to advance patient involvement and access. For more details, visit PatientsAsPartnersEU.com.
About the Conference Forum: The Conference Forum is a life science industry leader in organizing conferences, webinars, and digital content, driving efficient medicine development and patient access. Learn more at theconferenceforum.org.
SOURCE: EuropaWire
Pharma R&D and patient advocacy to discuss patient involvement in clinical research at Patients as Partners® Europe 2024
NEW YORK, NY, United States, 12-Mar-2024 — /EPR HEALTHCARE NEWS/ — The Conference Forum today announced the launch of the 8th annual Patients as Partners® Europe meeting, taking place May 14-15, 2024, at Plaisterers’ Hall, in London, England. Patients as Partners® Europe offers an unparalleled opportunity to hear from pharma R&D and patient advocacy on how patient involvement gets done to drive greater efficiencies and inclusivity in clinical research with better outcomes.
“Patients as Partners Europe brings advocates and sponsors together in a unique forum focused on improving collaborations that result in meaningful impact for patients,†said 2024 co-chair, Victoria DiBiaso, MPH, Global Head, Patient Informed Development & Health Value Translation, Sanofi.
The 2024 keynotes and featured speakers include:
- Patient Advocate Keynote Derek Stewart, OBE on understanding mental health support in clinical research
- Patient Advocate Keynote Sarah Zenner Dolan, a former biotech executive on navigating clinical trials as a patient
- GSK’s Andrew Garvey, Global Patient Advocacy Lead, on what has been working, and what has not, in GSK’s effort to advance patient engagement in clinical trials
- Pfizer’s Patrick Gallogly, Medical Advisor, Pfizer, on the learnings generated from an industry-first LGBTQ+ oncology advisory board
- Lundbeck’s Anders Lassen, Senior Director, Patient Insights, on using patient-centric integrated evidence approaches to inform drug development decisions
- Boehringer Ingelheim’s Annie Gilbert, Global Patient Advocacy Lead, on the pilot program bridging the communication gap by sending trial updates to patients
- Ipsen’s Oleksandr Gorbenko,​​​​​​​ Global Patient Affairs Director, Neurosciences, on how Ipsen partnered with patient advocacy to create robust patient experience mapping
- AstraZeneca’s Lisa Kerr, Senior Director, R&D Patient Science, on creating a measurement strategy and business investment case to scale patient-centric R&D approaches
- Astellas’ Stephen Head, Senior Director, Patient Partnerships, on instilling conscious awareness of the patient into everyday work practices
- Prostate Cancer Research’s David James, Director of Patient Projects, on demystifying and diversifying clinical trials to engage underserved communities
“Patients as Partners® Europe presents case examples on how patient involvement in clinical research can accelerate medicine development, how it can improve better access, inclusivity and diversity,†said Valerie Bowling, Executive Director.
Key topics to be addressed include:
- ​​​​The future of patient-informed research
- Understanding patient preferences in decentralized clinical trials
- Advancing health equity and diversifying clinical trials to engage underserved communities
- The patient experience data landscape and returning patient data
- Turning patient insights into action
- Mapping and measuring patient engagement
- Patient burden-reducing solutions and technologies
- Regulatory requirements and patient engagement in drug development
The 2024 meeting is co-chaired by ​​​​​​​Sanofi’s Victoria DiBiaso, MPH, Global Head, Patient Informed Development & Health Value Translation; Patient Advocate, ​​​​​​​​​​​​​​Alfred Samuels; and Parexel’s Rosamund Round, VP, Patient Engagement.
About Patients as Partners® Europe:
Patients as Partners® Europe is co-produced with patients, industry, academia, government and nonprofit organizations to establish a well-rounded program that addresses the needs of all stakeholders seeking to implement and advance patient involvement, access and diversity across the entire clinical development continuum. To learn more about Patients as Partners and access the full agenda, visit PatientsAsPartnersEU.com.
About the Conference Forum:
The Conference Forum is a life science industry research, conference development and marketing firm. The company brings the full spectrum of executives together to share ideas and information on how to advance efficient medicine development and delivery, patient diversity and access. They produce trusted conferences, webinars, podcasts, digital editorial and provide marketing services. To learn more, visit theconferenceforum.org.
SOURCE: EuropaWire
IQ Biozoom, a medtech startup, develops non-invasive home diagnostics to measure biomarker levels in saliva with unmatched accuracy
WARSAW, 13-Nov-2023 — /EPR HEALTHCARE NEWS/ — IQ Biozoom, a medical technology startup focusing on developing non-invasive home diagnostics, is set to make non-invasive home testing more convenient with their innovative technology that allows for the possibility to measure the concentration of biochemical substances in liquid analytes such as saliva, sweat, urine or tears. More people around the world will be empowered to monitor the course of various diseases from the comfort of their own home with laboratory precision using saliva analysis. Currently,  non-invasive home tests measuring glucose and lactate levels in saliva and, in the near future, hormones or CRP proteins, are being developed as part of the technology development.
IQ Biozoom’s technology includes a sophisticated biosensor system that integrates inkjet printing technology with advanced semiconductors, specifically thin-film transistors. Although not yet in commercial use, it allows the concentration of a selected biomarker to be determined based on contact between a disposable test strip and a body fluid such as saliva. According to analyses, when measuring glucose concentration in saliva, IQ Biozoom’s cutting-edge technology is up to ten times more accurate than current market standards. This means that even very low concentrations of selected biomarkers in the analyte can be determined with laboratory precision. This can be particularly relevant in those cases where the physiological concentration of a selected substance is very low, or where the mere presence of a biomarker in an analyte is indicative of a health disorder.
Most current blood glucose meters need a finger prick which is usually uncomfortable for patients. IQ Biozoom’s technology enables biomarker levels to be tested with laboratory precision based on saliva analysis, making it a completely non-invasive and painless method.
The solution will enable people with, for example, metabolic and endocrine disorders, to monitor their health and manage their disease, as well as take appropriate preventive care. The devices being developed based on IQ Biozoom’s technology are intended to be used for self-contained, non-invasive home diagnostics, allowing for precise laboratory-standard results.
Monitoring biomarkers and analysing their data retrospectively can help to select the right therapy for a patient. By monitoring biomarkers before, during and after therapy, it is possible to assess whether a patient is responding to treatment. If biomarker levels improve, this may indicate that the therapies are working. If there is no improvement, the therapy may need to be changed. Retrospective analysis of biomarker data can help to personalise therapy, tailoring it to the individual patient. The technology also has applications in personalised sports medicine, particularly in the context of physical performance assessment, training planning and training unit selection. Different athletes may have different lactate threshold which means their training plans should be tailored to their individual body characteristics. Testing lactate levels allows for precise individualisation of training.
Currently, invasive glucometers are the devices of choice for glucose level monitoring. According to the Polish Family with Diabetes report, published by the Association for Diabetes Education SED – 84% of people with type 1 diabetes would prefer to use non-invasive glucose monitoring methods, while 60% skip part of their daily testing due to the need for fingertip pricks. In addition to the discomfort felt, the disruption of the skin increases the risk of an infection. In 2021, IQ Biozoom started developing their innovative technology to non-invasively monitor glucose and lactate levels. Other biomarkers such as cortisol or CRP proteins in the body are in the R&D pipeline.
In the third quarter of 2023, IQ Biozoom obtained funding from the VC Link fund, which supports Polish companies developing breakthrough technologies with global potential. But the seed round is not closed yet with the startup seeking further investment to implement the technology and go to market.
The company expects an even faster growth rate in the near future. The home diagnostics market is growing at 5.6% year-to-year and is expected to exceed $13.6 million by 2033, according to FutureMarketInsight.com.
About IQ BIOZOOM
IQ BIOZOOM Ltd (https://iqbiozoom.com/) develops technology for non-invasive home diagnostics. The company is working on innovative devices for measuring the concentration of biochemical substances in saliva: glucose, lactates, and hormones, to enable people with metabolic and endocrine disorders to monitor their health and manage their disease, as well as applying appropriate prevention. IQ Biozoom technology will be designed for home self-testing. In the near future, the company intends to focus on expanding the technology usage to measure the levels of other substances important for diabetics (diabetes package) and woman (hormone package) ‒ in saliva.
SOURCE: EuropaWire
A groundbreaking paper: Climate change is a key driver of infectious disease outbreaks in Europe

BRUSSELS, 5-Sep-2023 — /EPR HEALTHCARE NEWS/ — The Lancet Regional Health – Europe journal has published a scientific paper on 7 August 2023 authored by a distinguished team of international researchers as part of their work in the IDAlert project. Titled “Decision-Support Tools to Build Climate Resilience Against Emerging Infectious Diseases in Europe and Beyond“, this paper introduces a transformative approach to tackle the emergence and transmission of climate-sensitive infectious diseases in Europe, informing cross-sectoral policy while improving the long-term climate resilience of health systems to infectious disease risks.
Climate change is one of several drivers of recurrent outbreaks and geographical range expansion of infectious diseases in Europe. This paper proposes a collaborative approach to develop policy-relevant indicators and decision-support tools. These tools are designed to comprehensively track and anticipate climate-induced disease risks across various domains, including environmental hazard, exposure patterns, and vulnerability factors. With a keen focus on the interconnectedness of animals, humans, and the environment, the framework promises a holistic perspective to address this multifaceted challenge.
The lead author and IDAlert project coordinator Joacim Rocklöv highlighted, “Our decision-support tools offer a multi-dimensional perspective that transcends traditional silos. By examining the nexus of animals, humans, and the environment, we’re unlocking a more comprehensive understanding of disease dynamics a pre-requisite for more timely and effective outbreak preparedness.â€
The heart of this novel framework lies in the co-production of early warning and response systems with stakeholders and end-users, as well as tailored tools to assess the costs and benefits associated with climate adaptation and mitigation strategies across diverse sectors. By fostering greater resilience within regional and local health systems, the framework aims to strengthen Europe’s capacity to respond to health crises, even in the face of changing environmental conditions.
As part of its approach, the IDAlert project will integrate multi-level engagement, innovative methodologies, and novel data streams, and tap into locally generated intelligence and empirical insights through case studies. This strategy empowers experts to quantify the effects of climate-induced disease threats in areas undergoing rapid urban transformation and contending with heterogeneous health risks. The ultimate aspiration is to bridge the gap between knowledge and action, delivering an unparalleled integrated One Health—Climate Risk framework that will empower policymakers, healthcare professionals, and communities to mitigate risks and bolster resilience.
About The Lancet Regional Health – Europe Journal:
The Lancet Regional Health – Europe Journal is a prestigious journal renowned for publishing groundbreaking research on health challenges worldwide. It aims to promote the advancement of the research agenda, clinical practice and health policy in Europe with the goal of improving health outcomes for all people regionally and globally.
About the IDAlert project:
IDAlert – Infectious Disease decision-support tools and Alert systems to build climate Resilience to emerging health Threats – officially started on 1 June 2022 is a € 9.18 million project and lasts for five years. The project is funded by the European Commission under the Horizon Europe programme with Grant Agreement number 101057554. More information: www.idalertproject.eu
Note to Editors:
For more information about the paper or to request an interview with the researchers, please contact the IDAlert project: contact@idalertproject.eu.
The full paper, “Decision-Support Tools to Build Climate Resilience Against Emerging Infectious Diseases in Europe and Beyond,†is available on the Lancet Regional Health Journal website:
https://www.thelancet.com/journals/lanepe/article/PIIS2666-7762(23)00120-5/fulltext
SOURCE: EuropaWire
New AI-powered predictive algorithm offers unprecedented accuracy in diagnosing NASH and liver fibrosis staging
BOSTON, MA, United States, 18-Jul-2022 — /EPR HEALTHCARE NEWS/ — Metadeq Inc., a global NASH and metabolic diseases diagnostics company, today announced a breakthrough non-invasive blood test that utilizes two novel circulating proteins to accurately diagnose Non-alcoholic steato-hepatitis (NASH) and liver fibrosis, and can score the stages of both diseases, without the need for invasive liver biopsy. The study which details the new blood test – just revealed in Gut, a leading hepatology and gastroenterology journal published by BMJ – was led by King’s College London Professor Geltrude Mingrone, in collaboration with Metadeq.
Approximately one in four people worldwide, or approximately two billion people, have Non-alcoholic fatty liver disease (NAFLD) and therefore are at risk of developing NASH. NASH is the most severe form of NAFLD, a silent disease associated with fatty deposit build-up of the liver. NASH leads to advanced liver diseases such as liver fibrosis (scarring), cirrhosis, and liver cancer, and may ultimately result in death. The annual cost to the US healthcare system alone has been estimated to be over $100 billion per year.
Currently, the only way to diagnose NASH is through the standard of diagnosis, invasive liver biopsy, which is expensive and is associated with significant complications and discomfort. Until now, no reliable blood test has been developed for NASH, nor liver fibrosis. Most importantly, no available test can define the severity (stage), or monitor the progression, of either NASH or liver fibrosis.

Professor Geltrude Mingrone from the School of Cardiovascular and Metabolic Medicine & Sciences at King’s College London, who is one of the most widely cited and influential researchers globally for metabolic diseases and disorders, set out to discover a more accurate liquid biopsy test in support of patient care and in collaboration with Metadeq. The resulting study was published today in leading journal, Gut.
This study identified two novel protein biomarkers, PLIN2 and RAB14, to assist in the diagnosis of patients with NASH and/or liver fibrosis. The ability of these proteins to detect both diseases was tested in cohorts with either NASH and/or liver fibrosis that were confirmed with liver biopsy, the current standard of diagnosis.
“This blood test will allow researchers and clinicians to define the prevalence of NASH across populations, including children and adolescents, avoiding the need for invasive liver biopsy,†said Professor Mingrone. “Critically, it will allow care-givers to monitor the efficacy of NASH treatments over time, reducing screen failures and helping generate better drugs.â€
“Since HEPAR-QTM is the only diagnostic modality that can diagnose NASH and fibrosis staging we expect it to be instrumental in the development of new therapeutics for the management of patients,†said Frank Jaksch, Chairman of Metadeq. “We believe that HEPAR-QTM will advance for the benefit of patients the problem of regular screening, that has resulted in late drug failures during clinical trials, since it is now possible to accurately measure the severity of disease in a non-invasive manner.â€
The Metadeq predictive algorithm, which uses A.I., provided unprecedented sensitivity (88-95%), specificity (90-100%), and overall accuracy (92-93%) for NASH, and also has near-perfect sensitivity (99%-100%), specificity (90%-96%), and accuracy (98%-99%) for liver fibrosis.
The test results show that the proteins can provide rapid and cost-effective testing to combat the growing epidemic of NASH and liver fibrosis. This could be an invaluable tool in diagnosing and monitoring cases of liver diseases, allowing people to receive earlier treatment, from lifestyle adjustments to surgical and pharmacological interventions.
Currently no NASH drug has been approved by either the FDA or EMA, which may be a direct result of the lack of an accurate, reliable, and non-invasive test. More than 65% of patients who enroll in clinical trials for NASH-related therapies are found to be ineligible for the trial due to screen failure, which causes major monetary losses to companies developing and testing NASH drugs. The improved accuracy and ability to detect NASH staging will help in identifying and enrolling the appropriate people in clinical trials, speeding up the development of NASH drugs.
About Metadeq
Metadeq Corp. is a diagnostics company that provides innovative liquid biopsy solutions for liver and metabolic diseases. For the first time, Metadeq allows people to measure the whole spectrum of liver diseases – from NAFLD, to NASH, and liver fibrosis – with a blood test. www.metadeq.com
SOURCE: EuropaWire
Silencil Reviews: A Scam Supplement Or Does It Really Treat Tinnitus – Critical Silencil Review

NEW YORK, 2022-Mar-24 — /EPR HEALTHCARE NEWS/ — It is no news that tinnitus has been considered an incurable health condition ever since it was discovered. Hence, whereas doctors and clinicians usually prescribe antidepressants or anxiety-relieving medications to assist reduce the internal noise, there are no medications specifically designed to help put an end to it. But can Silencil really cure tinnitus? A YES OR A NO!
The fact that no one believes tinnitus has a cure stems from the story that some people have attempted numerous approaches in an attempt to get rid of their ringing in the ears without success. Nonetheless, recent advancements in medical science are taking us one step closer to finding a cure – brain inflammation as the cause of tinnitus. Fortunately, the Silencil team has rallied around this cause (brain inflammation), and they are determined to see it through.
MUST SEE: VISIT THE OFFICIAL WEBSITE TODAY FOR YOUR OWN SUPPLEMENT
According to the manufacturer, Silencil was designed to be almost your one and only true protection against tinnitus of all forms. It is a new nutritional supplement that is intended to reduce ringing in the ears as well as any associated symptoms that may occur as a result of the condition by reducing brain inflammation. Silencil is believed to really solve tinnitus by targeting the underlying problem. This Silencil Review is meant to explain everything about this supplement. Real-world customers and doctors have endorsed it as being tested, dependable, approved, effective, potent, legitimate and reliable.
What Is Silencil?
Silencil is a natural tinnitus supplement made up of 28 clinically approved plant extracts and vitamins that have been shown to be effective in treating tinnitus by reducing brain inflammation. It improves overall brain function, as well as strengthening the immune system and repairing any damage that has occurred, thereby curing tinnitus.

CLICK HERE TO BUY THIS NUTRITIONAL SUPPLEMENT FOR YOUR TINNITUS ON THE OFFICIAL WEBSITE
Silencil is a brain-health supplement / vitamin for tinnitus which works to restore your brain and body’s normal function, ultimately treating you of the condition. Simultaneously providing users with a natural blend of plant and vitamin extracts that work on nerve cells and brain tissue, Silencil’s primary purpose is to improve the user’s ability to hear in all ramifications. While Silencil does assist to decrease the ringing in the ears, it does so primarily through the reduction of inflammation and the re-establishment of neural connections inside the brain.
Truly, Silencil should not be seen as a miracle solution for curing tinnitus, and thus, it should not be expected to produce results overnight. It is rather a dietary supplement which is vested with totally lowering the symptoms of tinnitus as a whole. It accomplishes relief to tinnitus sufferers by identifying and resolving the fundamental root of the problem known as brain inflammation. This is only possible if you have adhered to the manufacturer’s prescription and procedure.
According to general consensus, tinnitus is caused by an issue with one’s hearing. However, the inventor of Silencil believes that electrical misfires (inflammation) in the brain are responsible for a considerable percentage of the ringing in one’s ears and the Silencil pills are designed to address it as effectively as possible.
Is Silencil a legitimate treatment for tinnitus, or does it come with potentially serious side effects and allergies? Of course, this Silencil pill addresses and heals brain inflammation, which is the underlying cause of tinnitus in most cases. It is 100 percent legitimate, and the success stories that have been shared are genuine. Because the components in Silencil have all been approved by the FDA and professionally evaluated, there are no known negative side effects from using it. When the supplement is taken as directed, the pills are delivered directly to the brain, where they work to repair the damaged chain, alleviating the continual ringing sound in the ear.
This natural supplement is now widely acknowledged as one of the most effective natural remedies available for the treatment of tinnitus on a global scale. Millions of real clients all over the world have benefited from this superior supplement and have stated that the ringing or buzzing in their ears has been eliminated completely.
Via EPR Network
More Healthcare press releases
Kenneth Research: Increasing Concern for Mental Health Disorders to Drive Market Growth for Mental Health Software and Devices
NEW YORK, 21-Mar-2022 — /EPR HEALTHCARE NEWS/ — Every year, over 7 Million people are known to die by suicide, one of the major concerns of mental health, according to the statistics by the World Health Organization (WHO). Calculatedly, this comes to one person every 40 seconds. Moreover, in the year 2019, suicide accounted for 1.3% of the total number of deaths around the globe. On the other hand, mental health disorders, such as depression, affected 3.8% of the global population, or around 280 Million people. This included 5.0% adults and 5.7% aged adults above 60 years of age.
Kenneth Research recently released a report titled “Global Mental Health Software and Devices Market†which includes a detailed analysis of the market dynamics along with the impact of COVID-19 on the market growth during the forecast period, i.e., 2021-2030. The report also includes the regulatory and standard landscape, and in-depth research about the market growth across the five major regions, i.e., North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
In Europe, Austria (24 males, 5.6 females), Belgium (23 males, 8.6 females), France (22 males, 5.9 females), Finland (22.9 males, 7.6 females), Germany (17.3 males, 4.8 females), Sweden (17.7 males, 7.0 females), Netherlands (15.7 males, 7.0 females), has shown the highest number of death rates and self-harms per 100,000 inhabitants.
As COVID19 sweeps the global economies, Europe is one of the worst hit since 2020. As new COVID guidelines, lockdown and restriction comes into force, economies have reported elevated cases of anxiety and stress. Especially quarantine and its ill impacts have pushed levels of loneliness, depression, drug use, alcohol consumption, suicidal behavior and self-harm are pushed to another level.
Request for Research Report Sample-Â https://www.kennethresearch.com/report-details/mental-health-software-and-devices-market/10352350
Mental health is a major concern globally and with the rising prevalence of different types of such disorders in recent years, as well as with the growing awareness for these diseases by governmental bodies, the disease has been included in the list of 17 Sustainable Development Goals (SDGs) of the United Nations. Moreover, the situation for the disease has also gained massive focus, especially to treat the elderly population. This can be attributed primarily to the increasing elderly population count over the past few years, and the growing awareness for mental health and well-being amongst these age groups. In the other statistics by the WHO, the proportion of the population of age 60 years and over around the world is expected to double from 12% to 22% between 2015 and 2050. The statistics further stated that over 20% of these adults suffered from a mental or neurological disorder, and 6.6% of all disabilities amongst these population groups accounted for such disorders. Additionally, around 5% and 7% of these populations had been affected with dementia and depression respectively, which are the two most common mental and neurological disorders.
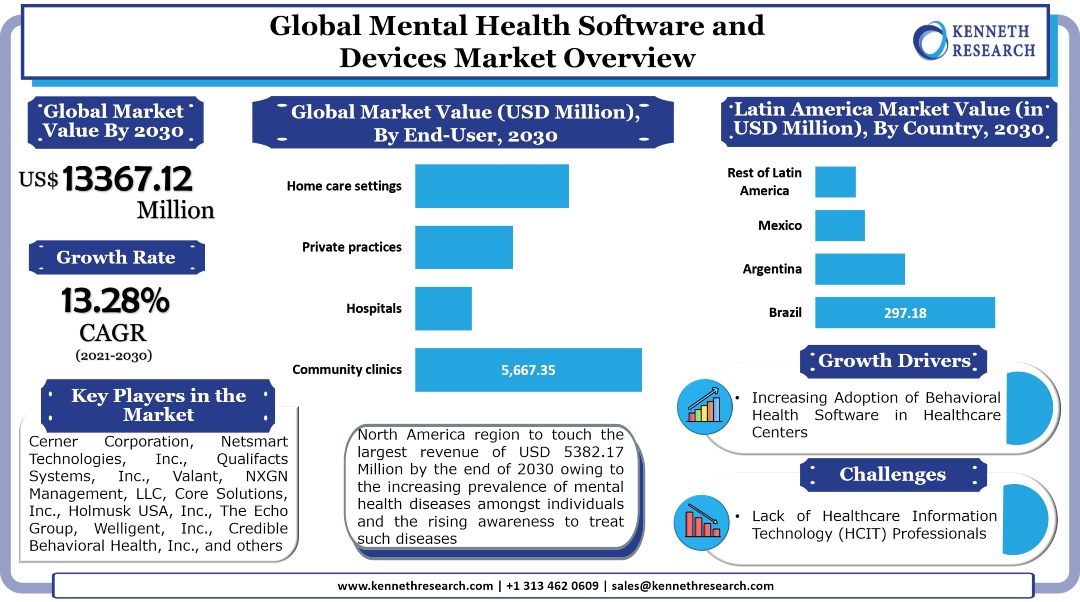
The global mental health software and devices market generated a revenue of USD 3883.94 Million in the year 2020 and is further expected to grow with a CAGR of 13.28% during the forecast period and touch a value of USD 13367.12 Million by the end of 2030. The growth of the market can primarily be attributed to the growing need for advanced patient monitoring and patient management applications amongst healthcare service providers for those treating mental disorders, backed by the surge in the number of mental health patients worldwide. Besides this, the increasing number of these patients is expected to drive the need for advanced devices which can help for early diagnosis and treatment, therefore contributing to the market growth in the coming years. For instance, the prevalence of dementia, which is known for deteriorating the memory, thinking, behavior, and the ability of the patients to perform everyday activities, is expected to increase to 82 Million by 2030, and further touch 152 Million by 2050, according to the statistics by the WHO. On the other hand, according to the statistics by United Nations Children’s Fund (UNICEF), in the year 2019, between the ages 10 and 14, 34,840,000 female adolescents and 44,647,000 male adolescents suffered from mental disorders. Moreover, between the ages 15 and 19, 41,712,000 female adolescents and 44,563,000 male adolescents had suffered from the same disorder. The surge in these cases is expected to boost the demand for diagnostic devices for the treatment of the disorder, and in turn, drive the market growth.
Request for Research Report Sample-Â https://www.kennethresearch.com/report-details/mental-health-software-and-devices-market/10352350
The growth of the global mental health software and devices market can also be attributed to the increasing number of government initiatives that focus on maintaining good mental health and well-being of the individuals of nations worldwide. For instance, in May 2013, during the 66th World Health Assembly, the Comprehensive Mental Health Action Plan 2013-2020 developed by the WHO was adopted by the Ministers of Health of the 194 Member States. This action plan was further extended until 2030 in the year 2019, during the 72nd World Health Assembly. On the other hand, according to the statistical report titled “Mental Health Atlas 2020â€, published by the WHO, 75% of the Member States had a stand-alone policy or plan for mental health. This was an increase from 68% in the year 2014.
Additionally, the statistics also stated that 57% of the Member States had a stand-alone mental health law. This was an increase from 51% in the year 2014. Besides this, since 2017, 46% of the WHO Member States had updated their health policy or plan, while 27% of them had updated their law regarding mental health.
The global mental health software and devices market is segmented on the basis of region into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. Out of the market in these regions, the market in North America generated the largest revenue of USD 1557.47 Million in the year 2020 and is further expected to touch USD 5382.17 Million by the end of 2030. One of the major factors anticipated to drive the growth of the market in the region is the increasing awareness for mental health and the rising need amongst healthcare service providers to improve their services to patients who are suffering from the disease. For instance, according to the statistics by the National Alliance on Mental Illness (NAMI), in the United States, in the year 2020, 1 in 5 U.S. adults experiences a mental illness, while 1 in 20 experienced a serious one, and 1 in 15 experienced both a substance use disorder and mental illness. In addition to this, 26.3 Million adults in the nation received virtual mental health services during the COVID-19 pandemic. Amongst those who received these services, 17.7 Million people experienced delays or cancellations in appointments, while 7.3 Million experienced delays in getting prescriptions. Moreover, 4.9 Million people were unable to access needed care. The market in the region is segmented by country into the United States and Canada. Out of the market in these countries, the market in the United States is expected to hold the largest revenue by the end of 2030 and also grow with the highest CAGR of 13.36% during the forecast period.
Request for Research Report Sample-Â https://www.kennethresearch.com/sample-request-10352350
On the other hand, the mental health software and devices market in Europe generated a revenue of USD 1265.71 Million in the year 2020 and is further expected to touch USD 4360.54 Million by the end of 2030, by growing with a CAGR of 13.31% during the forecast period. One of the major factors anticipated to drive the growth of the market in the region is the growing reforms in the diagnosis of mental health. For instance, on the 13th of May, 2019, the Parliamentary Assembly of the Council of Europe (PACE) on Social Affairs, Health, and Sustainable Development, had urged the member States of the Council of Europe to end coercive practices in mental health settings and further adopt a human rights-based approach which is respectful of medical ethics. The market growth in the region can also be attributed to the increasing government initiatives to improve behavioral health services and the surge in the adoption of electronic health records (EHR). The market in the region is further segmented by country into Germany, France, United Kingdom, Italy, Spain, Russia, Netherlands, and the Rest of Europe. Amongst the market in these countries, the market in France is expected to generate the largest revenue of USD 988.54 Million by the end of 2030 and further grow with a CAGR of 13.75% during the forecast period. Additionally, the market in the nation generated a revenue of USD 275.93 Million in the year 2020. Furthermore, the market in Germany registered the second-largest revenue of USD 249.35 Million in the year 2020 and is further expected to touch USD 911.35 Million by the end of 2030.
The study further incorporates Y-O-Y Growth, demand & supply and forecast future opportunity in North America (U.S., Canada), Europe (U.K., Germany, France, Italy, Spain, Hungary, Belgium, Netherlands & Luxembourg, NORDIC [Finland, Sweden, Norway, Denmark], Poland, Turkey, Russia, Rest of Europe), Latin America (Brazil, Mexico, Argentina, Rest of Latin America), Asia-Pacific (China, India, Japan, South Korea, Indonesia, Singapore, Malaysia, Australia, New Zealand, Rest of Asia-Pacific), Middle East and Africa (Israel, GCC [Saudi Arabia, UAE, Bahrain, Kuwait, Qatar, Oman], North Africa, South Africa, Rest of Middle East and Africa).
The global mental health software and devices market is segmented by delivery model into subscription model and ownership model. Amongst these segments, the subscription model segment is projected to garner the largest revenue of USD 2442.34 Million by the end of 2022. Further, the segment is also expected to grow with the highest CAGR of 11.82% during the forecast period. In North America, the segment is expected to garner the highest market share by the end of 2030, while in Europe, the segment is projected to garner the largest revenue of USD 762.37 Million by the end of 2022.
The global mental health software and devices market is further segmented by end user into community clinics, hospitals, private practices, and home care settings. Among these segments, the community clinics segment registered the largest revenue of USD 1644.62 Million in the year 2020 and is further expected to touch a revenue of USD 5667.35 Million by the end of 2030. In North America, the segment generated the largest revenue of USD 660.52 Million in the year 2020 and is further projected to reach USD 2285.27 Million by the end of 2030, by growing with a CAGR of 13.35% during the forecast period. On the other hand, in the Asia Pacific, the segment is anticipated to garner the largest revenue of USD 1124.71 Million by the end of 2030, up from a revenue of USD 317.70 Million in the year 2020. Moreover, in Japan, the segment is expected to garner the largest revenue of USD 244.73 Million and further grow with a CAGR of 14.34% during the forecast period.
The global mental health software and devices market is also segmented on the basis of component, functionality, and mode of access.
Global Mental Health Software and Devices Market, Segmentation by Component
- Software
o Integrated Software
o Standalone Software - Devices
Global Mental Health Software and Devices Market, Segmentation by Functionality
- Clinical Functionality
o Electronic Health Records (EHR)
o Clinical Decision Support (CDS)
o Care Plans/Health Management
o E-Prescribing
o Telehealth - Administrative Functionality
o Patient/Client Scheduling
o Document/Image Management
o Case Management
o Workforce Management
o Business Intelligence - Financial Functionality
o Revenue Cycle Management
o Managed Care
o Accounts Payable/General Ledger
Global Mental Health Software and Devices Market, Segmentation by Mode of Access
- Desktops/Laptops
- Tablets/Smartphone
Some of the prominent industry leaders in the global mental health software and devices market that are included in our report are Cerner Corporation, Netsmart Technologies, Inc., Qualifacts Systems, Inc., Valant, NXGN Management, LLC, Core Solutions, Inc., Holmusk USA, Inc., The Echo Group, Welligent, Inc., Credible Behavioral Health, Inc., IBM, and others.
Request for Research Report Sample-Â https://www.kennethresearch.com/sample-request-10352350
SOURCE: EuropaWire
Exopharm: exosomes seen as an alternative to lipid nanoparticles (LNPs) and adeno-associated viral vectors (#AAVs) for targeted multi-dose delivery of modern medicines and vaccines

MELBOURNE, Australia, 3-Feb-2022 — /EPR HEALTHCARE NEWS/ — Australian technologist Dr Ian Dixon founded Exopharm in 2013 with the vision to harness the unique properties of exosomes as a new class of medicine. Dixon’s decisive early entry into exosome therapeutics is now bearing fruits.
Less than 10 years after its founding, Exopharm possesses a granted US patent for its key exosome manufacturing technology, LEAP™, and has run successful first-in-human clinical trials demonstrating the safety of its exosome products.
Exosomes have the potential to revolutionise medicine in many areas, including delivery of gene therapies into cell nucleus for rare and common genetic disorders, improving therapeutic window of small and large molecules by cell targeting, and by selective cellular delivery of next-generation DNA/RNA products.
In 2022, excitement around exosomes has converged on exosomes as an alternative to lipid nanoparticles (LNPs) and adeno-associated viral vectors (AAVs) for targeted multi-dose delivery of modern medicines and vaccines. Drug delivery accounts for a significant slice of the global pharmaceutical industry spend –around US$160b p.a. and growing strongly – and exosomes represent nature’s way to deliver a bioactive cargo into cells efficiently and safely.
Primed for growth
Dixon’s establishment of a specialist exosome medicines company has been followed by US based Codiak Biociences in 2015 (established by Arch Ventures) and Oxford based Evox Therapeutics in 2016. Today, only two dedicated exosome companies are listed on the public markets: Codiak (NASDAQ: CDAK) with a market cap of around US$200m; and Exopharm (ASX: EX1) with a market cap of around A$73m at the time of writing this article.
The field of exosome medicines is attracting the attention of financiers and pharmaceutical industry leaders. In late 2021, multinational pharmaceutical manufacturing services company Lonza jumped into exosome manufacture, a sign that they see big things over the horizon.
Over the past 9 years, Exopharm has grown and evolved but remains true to Dixon’s vision of exosome medicine development. The company now employs around 40 people most in its Melbourne, Australia, facility. Exopharm has built a toolbox of exosome technologies and knowhow which, in addition to the LEAP™ scalable exosome manufacturing platform, includes exosome analytical tools (Exoria) and technologies to load exosomes with a specific therapeutic cargo (LOAD) and target exosomes to select cells (EVPS).
Our business development team is attracting the attention of similar innovative biopharmaceutical and biomanufacturing companies, and validating the Exopharm technologies via collaboration research agreements.
Exopharm is not simply a technology company, however. Exopharm is pursuing a pipeline driven platform technology strategy, on the way to generating a revenue stream from technology out-licensing while developing its own exosome medicines for selected markets. More news on its pipeline products is expected to be ready for release later in CY ’22.
The Exopharm team has grown and changed to becoming more international in focus, improving on messaging and communications with the wider pharmaceutical industry. In 2021 Dr Jenn King joined the Board of Exopharm, bringing a wealth of big pharma and US biotech experience with her.
In 2022 we look to engage with more experts, as members of our advisory panels, as consultants, executives or Key Opinion Leaders. Please reach out to us if you want to be part of our vision.
At the company’s 10-year anniversary in late 2023, Exopharm will be a key part of the exosome medicines revolution.
SOURCE: EuropaWire
EU Patients as Partners in Medicines Development conference to be held virtually on Jan 24-25, 2022

(PRESS RELEASE) New York, NY, United States, 5-Jan-2021 — /EPR HEALTHCARE NEWS/ — The Conference Forum announced the launch of the 6th annual EU Patients as Partners in Medicines Development conference to take place virtually on January 24-25th, 2022 GMT time.
​​This annual event, the first of its kind in Europe, will feature successful collaborations between patients and pharma where patient input directly impacted aspects of how a pharmaceutical company either designed or conducted a clinical trial.
Of the 20 sessions, half include a patient or patient advocate. This two-day program will demonstrate how patient-led research can improve the way pharma conducts clinical trials.
Examples include UCB developing new assessment tools to measure the effectiveness of novel treatments for Parkinson’s disease, changes in the design and implementation of an MS trial for greater patient engagement, and the development of a new toolkit to change the way patients are compensated for their contributions in clinical research.
“Patients have become more empowered,†says co-chair Michaela Dinboeck, Head, Center of Excellence for Patient Engagement at Novartis. “So patient engagement must be integrated into the DNA of how medicines are developed, approved, and used. Patients as Partners Europe will provide Pharma with valuable insights on how to achieve this.â€
Participating pharma companies include Novartis, Pfizer, Merck KGaA, EMD Serono, Janssen, Johnson & Johnson, GSK, Ipsen, UCB, Takeda, AbbVie, Noema Pharma, Gilead Sciences, Regeneron, and Pierre Fabre.
Other topics to be addressed include: Scaling patient engagement activities company-wide, engaging patients in early clinical development, leveraging social media to gain patient insights, diversity and inclusion in research and updates on patient engagement activities in Eastern Europe.
To learn more, please visit Patients as Partners Europe.
SOURCE: EuropaWire
Origami and citoQualis will work together to help Medical Device startups get products to global markets faster

KFAR SABA, Israel, 9-Nov-2021 — /EPR HEALTHCARE NEWS/ — Origami, a leading provider of no-code business process automation software for Medical Device and other industries, and citoQualis, a regulatory and business advisory leader for EU Medical Device companies, are announcing a partnership to help Medical Device startups get products to global markets faster.
Medical Device startups face unique pressures due to numerous country-specific regulations. This new partnership will help companies efficiently manage their regulatory approval processes, and enable rapid business growth.
Origami’s medical device customers operate around the world. With recent EU regulatory updates, companies are seeking strategic regulatory and business guidance to help them get and maintain product approval in that geography. The new partnership will provide Origami customers with access to citoQualis expertise in business and complex regulatory landscapes.
The citoQualis team points out that Medical Device startups have global workforces that “expect to access their systems from wherever they’re located, and to use those systems to collaborate internally and with citoQualis†says Mr. Wolfgang Werner, CEO of citoQualis.
According to Mr. Werner, “As advisors, it’s our job to identify opportunities and share our insights. For startups, the faster a product moves from idea to production, the better. With Origami’s flexibility, we are able to give our clients a software option that works for them now and as they scale.â€
Germany and other EU countries have strong medical device startup ecosystems, which citoQualis is well connected to. Origami is excited to work with citoQualis and the startups they advise, noting how both companies focus their solutions and teams on helping customers succeed in complex environments.
Yaniv Shimony, CEO of Origami says “startup CEOs recognize Origami’s business value, in that they always have the solution their company needs. When you create a new and innovative medical device, using inflexible software point solutions slows your business down. With Origami, information is instantly available about any part of your business such as sales, quality, design, product approvals, manufacturing, equipment maintenance, purchasing, supplier compliance, invoicing, complaints and more.â€
Existing customers will be able to maintain and expand EU market access by working with local regulatory experts, facilitating hundreds of millions of Euros of revenue.
SOURCE: EuropaWire
URSAPHARM and CEBINA collaborating to repurpose the antihistamine drug azelastine hydrochloride as a potential anti-SARS-CoV-2 therapeutic

SAARBRÜCKEN / VIENNA, 17-Feb-2021 — /EPR HEALTHCARE NEWS/ — In the fight against the SARS-CoV-2 virus, a large number of pharmaceutical companies are looking for new approaches to combat the pandemic. In addition to vaccines, intensive research and development efforts around the world are focusing on identifying new anti-COVID therapeutics that includes re-evaluation of existing product portfolios with a view to re-purpose drugs that have antiviral properties.
With this goal in mind, URSAPHARM Arzneimittel GmbH, based in Saarbrücken, and the Vienna-based biotech company CEBINA GmbH (Central European Biotech Incubator and Accelerator) are collaborating to repurpose the antihistamine drug azelastine hydrochloride as a potential anti-SARS-CoV-2 therapeutic. Azelastine has been successfully used for many years in anti-allergic therapy.
Starting with a novel computational drug repurposing prediction method, developed and performed by Prof Dr Robert Konrat (University of Vienna), CEBINA selected already approved active substances for experimental testing in collaboration with Prof Dr. Ferenc Jakab (Virology National Laboratory, Szentágothai-Research-Center, University of Pécs, Hungary). This collaborative effort led to the identification of azelastine as a potent antiviral compound effective against SARS-CoV-2, a finding confirmed by now by several independent research groups. The potent anti-SARS-CoV-2 activity of azelastine was demonstrated in a reconstituted human nasal tissue model and was evident at a lower dose than present in commercially available nasal sprays [1].
A clinical trial has started on February 16th 2021 to verify whether the use of a nasal spray preparation containing azelastine can achieve a reduction in viral load in the the nasal cavity. The aim of the study is to show whether the anti-allergic medication Pollival® Nasal Spray, or a diluted version, can achieve a reduction in the viral load, therefore reducing transmission and positively influencing the course of the disease.
If positive results of the running clinical studies are demonstrated, URSAPHARM Arzneimittel GmbH and CEBINA GmbH believe this will be an important new approach to control viral spread and contain the current pandemic.
SOURCE: EuropaWire
Code18 Interactive Launches New Websites for Balmex and Balmex Adult

New York, NY, 2021-Jan-20 — /EPR HEALTHCARE NEWS/ — New York digital agency Code18 Interactive gives two health and wellness brands—Balmex Complete Protection and Balmex AdultAdvantage—custom websites that will provide peace of mind and relief for everyone from baby to grandpa. Both sites are fully responsive and built on the WordPress platform.
Code18 Interactive, a boutique digital agency based in New York City, announces the launch of new health and wellness websites for Balmex Complete Protectin and Balmex AdultAdvantage. Both are fully custom and fully responsive WordPress websites – https://balmex.com helps elevate and refresh a legacy brand while providing helpful diaper rash solutions and peace of mind for new parents, and https://balmexadult.com showcases over-the-counter relief for adults who experience skin irritation and chafing caused by incontinence or light bladder leakage. Both sites also feature e-commerce capabilities powered by WooCommerce, enabling the brands to offer Direct-to-Consumer sales for the first time.
The Balmex Complete Protection and Balmex AdultAdvantage brands are both part of the Randob Labs portfolio. Randob Labs is a private, family-held company that specializes in revitalizing and maintaining heritage brands on the OTC healthcare space. “Collaborating and working with Randob Labs has been an exciting adventure. Both Balmex and Balmex Adult provide big relief from common, irritating issues,†says Code18 Interactive founder Steve Pilon. “And providing relief and solutions to everyday problems is exactly what Randob Labs is all about. We’re thrilled to be a part of this process.â€
As a well-established presence in the baby aisle, Balmex Complete Protection offers pediatrician-recommended diaper rash cream and multi-purpose healing ointment to help protect, soothe and heal baby’s delicate skin. “Many health and wellness websites only talk about what their product can do for consumers. However, Balmex is all about providing fast relief and reliable, proven solutions. Therefore, the website needed to clearly explain the causes of diaper rash and skin irritation, as well as provide simple steps parents can take to help prevent diaper rash in the first place,†says Pilon. “The brand’s willingness to go the extra mile for new parents is just one of the many reasons parents have been trusting Balmex for over 50 years.â€
Clinically proven Balmex AdultAdvantage offers the same key benefits—a cream that protects, soothes and heals irritated and chafed skin—as its 50-year-old sibling brand. Available as a standalone skin relief cream or as part of an innovative on-the-go, 3-in-1 kit, Balmex AdultAdvantage helps older adults live life more comfortably. “For many older adults incontinence or bladder leakage controls their life, and unfortunately, many people just accept this fate,†says Pilon. “With the Balmex Adult website we really focused on awareness—awareness of this fantastic product, awareness of the benefits, and mostly, awareness that you can take back control of your life with Balmex AdultAdvantage.â€
Code18 Interactive is a New York WordPress Agency that caters primarily to national consumer and media brands. The agency specializes in the design and development of enterprise-level WordPress and Magento 2 websites. They have the pleasure of working with many wonderful brands, including: GoGo squeeZ, Boursin Cheese, Public Radio International, The Laughing Cow, Grove Atlantic Publishing, C.O. Bigelow and many others.
For more information, please visit http://www.code18.com
Media contact:
Steve Pilon
Code18 Interactive
175 Varick Street, 8th Floor
New York, NY 10014 USA
Phone: 404-585-2725
https://code18.com
Via EPR Network
More Healthcare press releases
Palette Life Sciences unveils 4 international sites for its Deflux online presence

SANTA BARBARA, CALIF. / STOCKHOLM, 27-Oct-2020 — /EPR HEALTHCARE NEWS/ — Palette Life Sciences, a fully integrated global life sciences company dedicated to improving patient outcomes, today introduced valuable new online resources for healthcare providers in France, Germany, Italy and Spain. By expanding its Deflux online presence to international audiences, Palette Life Sciences demonstrates its commitment to supporting the global paediatric community.
Deflux.fr is a French-language resource for healthcare providers in France. Deflux.com/de is a German-language site serving practitioners in Germany. Deflux.com/es and Deflux.it are English-language sites serving physicians in Spain and Italy respectively. These new sites offer best-in-class online resources, important information and education about VUR, including symptoms, causes, risk factors and treatment options, such as Deflux®. All of the European sites provide physicians with access to the global Deflux Learning Center, a leading education platform that features information and training for paediatric urologists and their peers who treat VUR. The Deflux Learning Center includes clinical articles, educational videos, webinars, brochures, downloadable tools and The PedSpace podcast series, created for and presented by paediatric urology healthcare professionals.
The Deflux Learning Center for each respective country also provides downloadable educational materials for parents and caregivers managing children with vesicoureteral reflux (VUR).
“My personal long-term experience with Deflux has proven its effectiveness and safety in reducing VUR and preventing UTI recurrence in patients who fail medical management, thus avoiding use of long-term antibiotics. It is my hope that the new website initiative, which includes helpful resources for physicians, will help raise awareness of this treatment option in patients with primary VUR, troubled by recurrent febrile UTIs,†said Dr. Marie-Klaire Farrugia, Consultant Paediatric Urologist and Service Lead for Paediatric Surgery at Chelsea & Westminster and Imperial College Hospitals.
“We are excited to offer these new online resources to the healthcare community in France, Germany, Italy and Spain,†said Rich Low, Head of Marketing at Palette Life Sciences. “We are confident they will benefit the global paediatric urology ecosystem and serve as valuable resources by providing critical information and access to educational resources through the Deflux Learning Center.â€
SOURCE: EuropaWire
LIDBREE™ receives marketing authorization in the United Kingdom
SANTA BARBARA, CALIF., U.S.A. / BUDAPEST / STOCKHOLM, 5-Aug-2020 — /EPR HEALTHCARE NEWS/ — Palette Life Sciences AB (‘Palette’) with Gedeon Richter Plc. (‘Richter’), today announced that as a part of a decentralised regulatory procedure including thirty European Union Economic Area member states, Palette and Richter received national marketing authorization for LIDBREE™ in the United Kingdom (UK). The product is a novel, proprietary thermo gelling intrauterine formulation that can provide significant pain relief during common gynecological procedures.

LIDBREE’s marketing authorization is based on compelling data from clinical trials and investigations of sensory innervation in the reproductive area. In a pivotal clinical trial, LIDBREE treatment was associated with a significant reduction of pain and discomfort in women undergoing gynecological interventions.¹
“Our research group has detected pain nerves superficially throughout the uterus, which enable the use of local pain relief in many gynecological procedures such as insertion of an intrauterine device and sampling biopsies from the cervix and/or from the uterus – procedures that are currently routinely performed today without pain relief. LIDBREE is a novel gel containing the local anesthetic lidocaine which adheres to the mucosal tissue upon application to provide excellent analgesic effectâ€, says Gunvor Ekman-Ordeberg, MD PhD Professor at Karolinska Institute and founder of the LIDBREE program. “The gratification of observing the culmination of over 40 years of research into clinical practice via a new, simple, and effective treatment is a great privilege to share with all of my colleagues.â€
In October 2017, Richter and Palette signed an agreement to commercialise LIDBREE, a novel innovative proprietary pain relief pharmaceutical formulation, in Europe and in certain other markets.
“We are very pleased with the approval of LIDBREE in the UK, it is a great accomplishment from everyone involved. This is an important milestone and allows us to address a significant unmet medical need in women’s health. We look forward to supporting LIDBREE’s product launch through our partner Gedeon Richter and pursuing additional markets, such as the U.S., in collaboration with future regional partners.†says Helena Jansson, Managing Director at Palette Life Sciences AB.
“We’ve been expecting this regulatory development and we are looking forward to successfully introduce LIDBREE to all of the territories covered by our agreement in order to further improve the quality of life of women experiencing pain during their gynaecological interventions,†stated Tibor Horváth, Commercial Director of Gedeon Richter Plc.
SOURCE: EuropaWire
Palette Life Sciences unveils an online resource aimed at paediatric urologists, parents and caregivers in the UK dealing with vesicoureteral reflux (VUR)

SANTA BARBARA, CALIF., U.S.A. / STOCKHOLM SWEDEN, 4-Aug-2020 — /EPR HEALTHCARE NEWS/ — Palette Life Sciences, a fully integrated global life sciences company dedicated to improving patient outcomes, today announced the launch of Deflux.com/UK as part of its global expansion. Deflux.com/UK is a best-in-class online resource developed specifically for healthcare practitioners in the United Kingdom, as well as parents and caregivers managing children with vesicoureteral reflux (VUR). VUR, sometimes referred to as urinary or bladder reflux, is a serious paediatric urinary condition in which urine flows from the bladder through one or both ureters toward the kidneys.
Deflux.com/UK features important information about VUR, including symptoms, causes, risk factors and treatment options, such as Deflux®. Comprised of hyaluronic acid (HA) and dextranomer polysaccharides to form a gel, Deflux is a minimally invasive biocomopatible treatment for VUR. Endoscopic treatment with Deflux takes about 15 minutes in a day procedure setting, and generally allows the child to go back to normal activities the next day.
Deflux.com/UK also provides physicians with access to the Deflux Learning Center, a leading global education platform that features information and training for paediatric urologists and their peers who treat VUR as well as other paediatric urology conditions. The Deflux Learning Center includes clinical articles, educational videos and webinars, brochures, downloadable tools and The PedSpace podcast series, created for and presented by paediatric urology healthcare professionals.
“Deflux is an effective, minimally-invasive treatment option that many caregivers simply aren’t aware of,†said Dr. Marie-Klaire Farrugia, Consultant Paediatric Urologist and Clinical Director for Paediatric Surgery at Chelsea and Westminster Hospital. “My personal long-term experience with Deflux has proven its effectiveness and safety in reducing VUR and preventing UTI recurrence, thus avoiding use of long-term antibiotics. It is my hope that the new website initiative, which includes helpful resources for both physicians and caregivers, will help raise awareness and utilisation of this treatment option.â€
“The entire Palette commercial team is excited to launch Deflux.com/UK, which we believe will help elevate the standard of care for VUR patients in the UK,†said Rich Low, Head of Marketing, Palette Life Sciences. “Deflux.com/UK will serve as a truly valuable resource for caregivers, providing critical information to paediatric urology healthcare professionals through the Deflux Learning Center, and to caregivers, connecting them to highly-trained specialists who provide high quality care.â€
SOURCE: EuropaWire
Palette Life Sciences expands distribution network across Europe

STOCKHOLM, SWEDEN / SANTA BARBARA, CALIF., USA, 15-Jun-2020 — /EPR HEALTHCARE NEWS/ — Palette Life Sciences, a fully integrated global life sciences company dedicated to improving patient outcomes, today announced global distribution expansion for two of Palette’s products, Deflux and Solesta. The expansion provides commercial access to more than 20 countries through five distributors around the globe, while adding direct sales in four additional countries: Cyprus, Greece, Hungary and Portugal.
KEBOMED, a leading European distributor of medical devices and equipment, will distribute the products in France, Sweden, Norway, Finland, Denmark and Iceland. Stratarium Sarl, will commercialize the products in Germany, Austria, Belgium, Luxembourg, Netherlands and Switzerland leveraging their urology focused subsidiary, RA Distribution. CJ Medical, a UK-based distributor of innovative technologies specializing in surgical and medical devices, will distribute the products in Ireland and the United Kingdom. Grifols, one of the leading companies in the production and sale of plasma-derived medicines and a top distributor of a broad range of products within their country will cover Spain, and GP Dermal, a specialist with deep ties across urology and pediatric call points will serve as the distributor in Italy and Malta.
“This enhanced distribution network allows Palette to ensure our customer and patient needs are adequately met across Europe,†said James Leech, Head of Corporate Development at Palette Life Sciences. “We have selected strong partners with deep experience and relationships in the urology and pediatric setting who will help us serve the product and education needs of physicians and hospitals. Palette and its network of partners are making significant investments within local markets to improve physician access to Palette resources, best management and treatment practices, and renewed product training.â€
For more information or to place an order, please contact a Palette Life Sciences representative at eusales@palettelifesciences.com. Contact information for product orders or inquires by distributor are as follows:
KEBOMED: ordre@kebomed.dk
Stratarium Sarl: deflux-solesta@stratarium.com
CJ Medical: office@cjmedical.com
Grifols: jose.sagales@grifols.com
GP Dermal: luigi.galandini@gpdermal.com
SOURCE: EuropaWire
American Hemp Processing To Roll Out Mobile Extraction Units
Sacramento, California, 2019-Dec-10 — /EPR HEALTHCARE NEWS/ — American Hemp Processing (AHP) is excited to announce the design completion of their Mobile Hemp Extraction Unit (MEHU). This is a key step in AHP’s expansion to increase production to 400,000lbs of hemp per month that will be completed 1st quarter 2020. The mobility of the MEHU reduces overall processing costs, mitigates many of the risks associated to transportation, and increases transparency with farmers.
The MHEU units bring significant benefits to the farmer from both a financial and risk mitigation perspective. “Most hemp farms are not close to processing facilities. Transportation is a large cost and increases the risk of degradation and contamination. On top of that, farmers still have to deal with legal issues in states that aren’t familiar with hemp and mistake it for marijuana,†says Andrew Alvarez Co-Founder and Chief Technology Officer “Additionally, it increases transparency with farmers and helps educate them along the way to build synergistic relationshipsâ€.
“The design of the MHEU was Phase 2 of our strategy,†says Shick Park Co-Founder and Chief Operations Officer, “Phase 1 was developing a scalable extraction and remediation process enabling us to create high quality and compliant CBD oil, and Phase 3 is the roll-out of the mobile extraction units on to farms fall of 2020.â€
“This caps off a 12 month design process by our engineers and now we go to the assembly phase. These units will take approximately 120 days each to build, and we plan to initially build 3,†says Co-Founder and CEO Tom Richardson. “This has been a very rewarding journey for us, from meeting farmers all over the country, to government legislators, to fine tuning the extraction process, to meeting other processors and sharing ideas, we feel very confident about the Cannabinoid Sector business.â€
Via EPR Network
More Healthcare press releases
Parenteral Drug Association’s “ICH Q7 GMP for APIs” online training module is now endorsed by ICH

BERLIN, 22-Nov-2019 — /EPR HEALTHCARE NEWS/ — The Parenteral Drug Association (PDA) is proud to announce that its “ICH Q7 GMP for APIs†online training module is now endorsed by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use as an “ICH Recognised Training Programme.â€
The training module received the distinction after PDA demonstrated to ICH that it meets the twelve eligibility criteria for the program. Among these are its mission to serve as a neutral convening platform to engage expert stakeholders from industry, academia, advocacy groups, non-profits, and/or regulatory agencies on critical regulatory topics, and its promotion of regulatory convergence to accelerate innovation and improve healthcare globally.
The free “ICH Q7 GMP for APIs†training module introduces and improves understanding of the ICH quality guideline, Q7: Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients. The webinar is intended for pharmaceutical professionals working with APIs. More information on the webinar is available here: https://www.pda.org/global-event-calendar/ich-q7-gmp-for-apis.
SOURCE: EuropaWire
###


