
Category Archives: Business of Medicine


Myeloid Therapeutics rebrands and unveils an expanded strategy to move beyond its previous myeloid cell focus
- Proof-of-mechanism: Paired biopsies confirmed CAR+ immune cells infiltrating tumors, with immune remodeling and CD8 T cell recruitment.
- Safety and repeat dosing: Over 200 doses delivered with a consistent, manageable safety profile and no cumulative toxicities.
- Evidence of activity: CAR expression detected in circulating immune cells, with stable disease in several patients and a confirmed partial response on treatment for 16 months.
- MT-302 (TROP2; solid tumors): Dose escalation completed; tolerable safety profile.
- MT-303 (GPC3; hepatocellular carcinoma): Dose escalation ongoing.
- MT-304 (HER2; solid tumors): First patient expected Q4 2025; first-in-class multi-immune CAR engaging NK and myeloid cells.
- Retrotransposon-based in vivo CAR-T: First all-RNA product candidate with permanent CAR integration for B-cell depletion.
- Additional multi-lineage programs in preclinical development across oncology and immunology.
- Selective activation: Cell-specific receptors and LNPs for precise T, myeloid, and NK cell programming.
- Flexible durability: Transient or stable CAR expression, with permanent integration via RNA-based retrotransposon technology.
- Industry-leading RNA: Up to 8+ days of linear mRNA expression with no reactogenicity.
- Speed and scalability: Concept-to-clinic in <12 months with low-cost manufacturing.
EU Consortium Launches AI‑Driven “Digital Twin” Platform to Transform Aortic Surgery
 ITALY, 15-Jul-2025 — /EuropaWire/ — An EU‑funded consortium of Italian and French partners has unveiled PANDORA, a pioneering in‑silico platform designed to transform aortic aneurysm surgery, according to a recent press release announcement made on EuropaWire. Developed by LivGemini, RBF Morph, INSA and Université de Rennes (UnivREN) in collaboration with Rennes University Hospital, PANDORA creates patient‑specific “digital twins” of the aorta by combining AI‑driven image analysis, mesh‑morphing algorithms and high‑performance simulations.
ITALY, 15-Jul-2025 — /EuropaWire/ — An EU‑funded consortium of Italian and French partners has unveiled PANDORA, a pioneering in‑silico platform designed to transform aortic aneurysm surgery, according to a recent press release announcement made on EuropaWire. Developed by LivGemini, RBF Morph, INSA and Université de Rennes (UnivREN) in collaboration with Rennes University Hospital, PANDORA creates patient‑specific “digital twins” of the aorta by combining AI‑driven image analysis, mesh‑morphing algorithms and high‑performance simulations.
Surgeons can run virtual procedures on these personalized models to evaluate potential complications—such as graft kinking, impaired coronary perfusion and abnormal wall stresses—before entering the operating room. The goal is to guide prosthesis selection and surgical strategy with objective, data‑backed insights rather than relying solely on individual experience.
Offered under a B2B2B licensing model, the software will be integrated by medical‑device manufacturers into their planning systems for hospitals and clinics. Project coordinator Dr. Leonardo Geronzi, a Forbes “Top Under 30” in Science & Health, says PANDORA aims to shorten operating times, improve postoperative outcomes and reduce costs. After clinical validation at Rennes, LivGemini plans to industrialize the solution for broader market release later this year.
SOURCE: EuropaWire
9th Annual Patients as Partners® Europe Event to Focus on Advancing Patient Involvement in Clinical Research

NEW YORK, NY, United States, 11-Feb-2025 — /EuropaWire/ — The Conference Forum has announced the 9th annual Patients as Partners® Europe event, set for May 20-21, 2025, at the Royal National Hotel in London. This event provides a unique platform for pharma R&D professionals and patient advocates to discuss how patient involvement can reduce risks and inefficiencies in clinical research while advancing the development of new treatments.
Alfred Samuels, a patient advocate and co-chair for 2025, emphasized the importance of recognizing patients as experts in their conditions, which leads to more successful trials and better healthcare outcomes.
Keynote speakers include:
- Harry Verbunt, who will share his post-cancer clinical trial experience and suggestions for improving trial access.
- Peter DiBiaso, discussing the importance of placing patients at the center of research.
- Dr. Temi Olonisakin, offering patient perspectives on support in clinical research.
- Lea-Isabelle Proulx, PhD (Roche), on reducing patient burden through improved protocols.
- Beyza Klein (Johnson & Johnson), on embedding a patient engagement strategy.
- Dr. Catherine Coulouvrat (Sanofi), discussing how patient experience data informs product tolerability.
- Marisa Minetti (Chiesi Farmaceutici), on leveraging patient archetypes in protocol development.
- Gisela Linthorst (Azafaros), on involving the patient community in early clinical trials.
Pharma companies such as AstraZeneca, Boehringer-Ingelheim, GSK, and others will also present case studies on advancing patient partnerships.
The event will explore critical topics including patient-informed research, decentralized trials, increasing trial representation, and reducing patient burden through technology.
Patients as Partners® Europe is co-produced with patients, R&D, academia, and non-profit organizations to advance patient involvement and access. For more details, visit PatientsAsPartnersEU.com.
About the Conference Forum: The Conference Forum is a life science industry leader in organizing conferences, webinars, and digital content, driving efficient medicine development and patient access. Learn more at theconferenceforum.org.
SOURCE: EuropaWire
Pharma R&D and patient advocacy to discuss patient involvement in clinical research at Patients as Partners® Europe 2024
NEW YORK, NY, United States, 12-Mar-2024 — /EPR HEALTHCARE NEWS/ — The Conference Forum today announced the launch of the 8th annual Patients as Partners® Europe meeting, taking place May 14-15, 2024, at Plaisterers’ Hall, in London, England. Patients as Partners® Europe offers an unparalleled opportunity to hear from pharma R&D and patient advocacy on how patient involvement gets done to drive greater efficiencies and inclusivity in clinical research with better outcomes.
“Patients as Partners Europe brings advocates and sponsors together in a unique forum focused on improving collaborations that result in meaningful impact for patients,†said 2024 co-chair, Victoria DiBiaso, MPH, Global Head, Patient Informed Development & Health Value Translation, Sanofi.
The 2024 keynotes and featured speakers include:
- Patient Advocate Keynote Derek Stewart, OBE on understanding mental health support in clinical research
- Patient Advocate Keynote Sarah Zenner Dolan, a former biotech executive on navigating clinical trials as a patient
- GSK’s Andrew Garvey, Global Patient Advocacy Lead, on what has been working, and what has not, in GSK’s effort to advance patient engagement in clinical trials
- Pfizer’s Patrick Gallogly, Medical Advisor, Pfizer, on the learnings generated from an industry-first LGBTQ+ oncology advisory board
- Lundbeck’s Anders Lassen, Senior Director, Patient Insights, on using patient-centric integrated evidence approaches to inform drug development decisions
- Boehringer Ingelheim’s Annie Gilbert, Global Patient Advocacy Lead, on the pilot program bridging the communication gap by sending trial updates to patients
- Ipsen’s Oleksandr Gorbenko,​​​​​​​ Global Patient Affairs Director, Neurosciences, on how Ipsen partnered with patient advocacy to create robust patient experience mapping
- AstraZeneca’s Lisa Kerr, Senior Director, R&D Patient Science, on creating a measurement strategy and business investment case to scale patient-centric R&D approaches
- Astellas’ Stephen Head, Senior Director, Patient Partnerships, on instilling conscious awareness of the patient into everyday work practices
- Prostate Cancer Research’s David James, Director of Patient Projects, on demystifying and diversifying clinical trials to engage underserved communities
“Patients as Partners® Europe presents case examples on how patient involvement in clinical research can accelerate medicine development, how it can improve better access, inclusivity and diversity,†said Valerie Bowling, Executive Director.
Key topics to be addressed include:
- ​​​​The future of patient-informed research
- Understanding patient preferences in decentralized clinical trials
- Advancing health equity and diversifying clinical trials to engage underserved communities
- The patient experience data landscape and returning patient data
- Turning patient insights into action
- Mapping and measuring patient engagement
- Patient burden-reducing solutions and technologies
- Regulatory requirements and patient engagement in drug development
The 2024 meeting is co-chaired by ​​​​​​​Sanofi’s Victoria DiBiaso, MPH, Global Head, Patient Informed Development & Health Value Translation; Patient Advocate, ​​​​​​​​​​​​​​Alfred Samuels; and Parexel’s Rosamund Round, VP, Patient Engagement.
About Patients as Partners® Europe:
Patients as Partners® Europe is co-produced with patients, industry, academia, government and nonprofit organizations to establish a well-rounded program that addresses the needs of all stakeholders seeking to implement and advance patient involvement, access and diversity across the entire clinical development continuum. To learn more about Patients as Partners and access the full agenda, visit PatientsAsPartnersEU.com.
About the Conference Forum:
The Conference Forum is a life science industry research, conference development and marketing firm. The company brings the full spectrum of executives together to share ideas and information on how to advance efficient medicine development and delivery, patient diversity and access. They produce trusted conferences, webinars, podcasts, digital editorial and provide marketing services. To learn more, visit theconferenceforum.org.
SOURCE: EuropaWire
Novartis, Lundbeck, and GSK Among Pharmaceutical Companies to Present at Patients as Partners Europe 2023
NEW YORK, NY, United States, 13-Apr-2023 — /EPR HEALTHCARE NEWS/ — The Conference Forum announced that the 7th annual European Patients as Partners® in Clinical Research conference will return to London at the Thistle Hotel Marble Arch on June 12-13, 2023. The event will showcase the progress made by pharma R&D executives with input from patients on achieving greater representation in clinical research, building trust and engagement in communities, and building truly patient-centric R&D organizations.
“We are delighted to be back in person to offer an unparalleled opportunity to hear from pharma R&D and patient advocacy together on how patient involvement gets done to drive greater efficiencies in clinical research with better outcomes,†said Valerie Bowling, Executive Director, Patients as Partners in Clinical Research Europe.
A variety of pharmaceutical companies will report on the progress they’ve made engaging communities and partnering with patients. Sanofi will open with the evolution of patient-informed research and its impact on medicine development. Novartis will be speaking on the organization’s global approach to implementing systematic and consistent patient engagement across the medicine lifecycle with a focus on measurement.
Lundbeck will share how they are generating insights through engagement with patient communities and making patient input actionable for medicines development. CSL Behring will discuss how they are evaluating clinical research sites based on patient-focused principles.
Several companies, including Regeneron, the Center for BME Health and Sanofi, will discuss their progress in increasing diversity amongst underrepresented communities in clinical research. GSK will lead a session on understanding barriers to community engagement in clinical research.
Boehringer Ingelheim will discuss the company’s approach to engaging patients throughout the clinical trial and how to leverage those patient insights to improve the process. Alnylam Pharmaceuticals will share how the organization has developed a top-down approach to integrating the patient perspective.
The conference features incredible patient advocate representation, addressing the realities of underrepresented communities to better serve them. For the first time, Patients as Partners Europe will have a patient-led session on supporting mental health for patients in clinical research.
“I look forward to hearing from patients and industry colleagues about what matters most and what has the greatest impact when partnering with patients and their communities on our efforts to new therapeutic options available,†said Deirdre BeVard, SVP, R&D Strategic Operations, CSL Behring and Patients as Partners Europe co-chair.
Key topics for 2023 include:
- The evolution of patient informed research
- Increasing diversity in clinical research
- Patient access to research through digital innovation
- Understanding barriers to community engagement in clinical research
- Turning patient insights into action
- Navigating patient engagement and compliance
- Mapping and how to measure patient engagement
- Regulatory requirements and patient engagement in drug development
To learn more about the event, and a full overview of speakers, please click here (https://theconferenceforum.org/conferences/patients-as-partners-europe/overview/)
About Patients as Partners EU
Patients as Partners is co-produced with patients, industry, academia, government and nonprofit organizations to establish a well-rounded program that addresses the needs of all stakeholders seeking to implement and advance patient involvement across the entire clinical development continuum.
About the Conference Forum
The Conference Forum is a life science industry research firm that develops conferences primarily around how to get therapeutics to patients faster. They examine and challenge the complex ecosystem of drug development and delivery, bringing ideas together from a variety of sources to help advance clinical research with common goals that are patient-focused.
They currently offer conferences for pharma/biotech professionals including R&D leaders, CEOs, business development/licensing, medical affairs/safety, chief patient officers/advocates, clinical innovation champions, investors and drug delivery specialists. The company also publishes six newsletters and produces PharmaTalkRadio and virtual events.
SOURCE: EuropaWire

Cole & Van Note Announces Mon Health Data Breach Investigation

Oakland, California, USA, 2022-Mar-05 — /EPR HEALTHCARE NEWS/ — Cole & Van Note, a leading consumer rights law firm, announces today its investigation of Monongalia Health System, Inc. on behalf of its consumers/clients, arising out the company’s recent data breach. According to the company, the private information of a massive number of people may have been stolen in the hacking of its information network. It is currently unknown how many people have had their information used for criminal purposes.
If you received a notice of this alarming data breach and/or have transacted in any way with Monongalia Health System, Inc., your information may already be in the hands of cybercriminals, making your urgent attention to this situation very important.
Cole & Van Note is ready to discuss your options and can be contacted at (510) 891-9800, by email at sec@colevannote.com or through its website by clicking below:
Cole & Van Note has been successfully handling consumer and employee rights matters since 1992. The firm has recovered compensation for millions of individuals and stands ready to help you get paid for your losses.
Attorney Advertisement. Our previous results do not guarantee or predict a similar outcome.
Full Name: Scott Cole
Organization Name: Cole & Van Note
Phone: (510) 891-9800
Email Address: sec@colevannote.com
Facebook Page
LinkedIn Page

Cole & Van Note Announces SAC Health System Data Breach Investigation

Oakland, California, USA, 2022-Mar-05 — /EPR HEALTHCARE NEWS/ — Cole & Van Note, a leading consumer rights law firm, announces today its investigation of SAC Health System on behalf of its consumers/clients, arising out the company’s recent data breach. According to the company, the private information of a massive number of people may have been stolen in the hacking of its information network. It is currently unknown how many people have had their information used for criminal purposes.
If you received a notice of this alarming data breach and/or have transacted in any way with SAC Health System, your information may already be in the hands of cybercriminals, making your urgent attention to this situation very important.
Cole & Van Note is ready to discuss your options and can be contacted at (510) 891-9800, by email at sec@colevannote.com or through its website by clicking below:
Cole & Van Note has been successfully handling consumer and employee rights matters since 1992. The firm has recovered compensation for millions of individuals and stands ready to help you get paid for your losses.
Attorney Advertisement. Our previous results do not guarantee or predict a similar outcome.
Full Name: Scott Cole
Organization Name: Cole & Van Note
Phone: (510) 891-9800
Email Address: sec@colevannote.com
Facebook Page
LinkedIn Page
Via EPR Network
More Healthcare press releases

Cole & Van Note Announces Logan Health Medical Center Data Breach Investigation

Oakland, CA, USA, 2022-Mar-03 — /EPR HEALTHCARE NEWS/ — Cole & Van Note, a leading consumer rights law firm, announces today its investigation of Logan Health Medical Center on behalf of its consumers/clients, arising out the company’s recent data breach. According to the company, the private information of a massive number of people may have been stolen in the hacking of its information network. It is currently unknown how many people have had their information used for criminal purposes.
If you received a notice of this alarming data breach and/or have transacted in any way with , Logan Health Medical Center your information may already be in the hands of cybercriminals, making your urgent attention to this situation very important.
Cole & Van Note is ready to discuss your options and can be contacted at (510) 891-9800, by email at sec@colevannote.com or through its website by clicking below:
Cole & Van Note has been successfully handling consumer and employee rights matters since 1992. The firm has recovered compensation for millions of individuals and stands ready to help you get paid for your losses.
Attorney Advertisement. Our previous results do not guarantee or predict a similar outcome.
Full Name: Scott Cole
Organization Name: Cole & Van Note
Phone: (510) 891-9800
Email Address: sec@colevannote.com
Facebook Page
LinkedIn Page
Via EPR Network
More Healthcare press releases
EU Patients as Partners in Medicines Development conference to be held virtually on Jan 24-25, 2022

(PRESS RELEASE) New York, NY, United States, 5-Jan-2021 — /EPR HEALTHCARE NEWS/ — The Conference Forum announced the launch of the 6th annual EU Patients as Partners in Medicines Development conference to take place virtually on January 24-25th, 2022 GMT time.
​​This annual event, the first of its kind in Europe, will feature successful collaborations between patients and pharma where patient input directly impacted aspects of how a pharmaceutical company either designed or conducted a clinical trial.
Of the 20 sessions, half include a patient or patient advocate. This two-day program will demonstrate how patient-led research can improve the way pharma conducts clinical trials.
Examples include UCB developing new assessment tools to measure the effectiveness of novel treatments for Parkinson’s disease, changes in the design and implementation of an MS trial for greater patient engagement, and the development of a new toolkit to change the way patients are compensated for their contributions in clinical research.
“Patients have become more empowered,†says co-chair Michaela Dinboeck, Head, Center of Excellence for Patient Engagement at Novartis. “So patient engagement must be integrated into the DNA of how medicines are developed, approved, and used. Patients as Partners Europe will provide Pharma with valuable insights on how to achieve this.â€
Participating pharma companies include Novartis, Pfizer, Merck KGaA, EMD Serono, Janssen, Johnson & Johnson, GSK, Ipsen, UCB, Takeda, AbbVie, Noema Pharma, Gilead Sciences, Regeneron, and Pierre Fabre.
Other topics to be addressed include: Scaling patient engagement activities company-wide, engaging patients in early clinical development, leveraging social media to gain patient insights, diversity and inclusion in research and updates on patient engagement activities in Eastern Europe.
To learn more, please visit Patients as Partners Europe.
SOURCE: EuropaWire
Origami and citoQualis will work together to help Medical Device startups get products to global markets faster

KFAR SABA, Israel, 9-Nov-2021 — /EPR HEALTHCARE NEWS/ — Origami, a leading provider of no-code business process automation software for Medical Device and other industries, and citoQualis, a regulatory and business advisory leader for EU Medical Device companies, are announcing a partnership to help Medical Device startups get products to global markets faster.
Medical Device startups face unique pressures due to numerous country-specific regulations. This new partnership will help companies efficiently manage their regulatory approval processes, and enable rapid business growth.
Origami’s medical device customers operate around the world. With recent EU regulatory updates, companies are seeking strategic regulatory and business guidance to help them get and maintain product approval in that geography. The new partnership will provide Origami customers with access to citoQualis expertise in business and complex regulatory landscapes.
The citoQualis team points out that Medical Device startups have global workforces that “expect to access their systems from wherever they’re located, and to use those systems to collaborate internally and with citoQualis†says Mr. Wolfgang Werner, CEO of citoQualis.
According to Mr. Werner, “As advisors, it’s our job to identify opportunities and share our insights. For startups, the faster a product moves from idea to production, the better. With Origami’s flexibility, we are able to give our clients a software option that works for them now and as they scale.â€
Germany and other EU countries have strong medical device startup ecosystems, which citoQualis is well connected to. Origami is excited to work with citoQualis and the startups they advise, noting how both companies focus their solutions and teams on helping customers succeed in complex environments.
Yaniv Shimony, CEO of Origami says “startup CEOs recognize Origami’s business value, in that they always have the solution their company needs. When you create a new and innovative medical device, using inflexible software point solutions slows your business down. With Origami, information is instantly available about any part of your business such as sales, quality, design, product approvals, manufacturing, equipment maintenance, purchasing, supplier compliance, invoicing, complaints and more.â€
Existing customers will be able to maintain and expand EU market access by working with local regulatory experts, facilitating hundreds of millions of Euros of revenue.
SOURCE: EuropaWire
University of Freiburg spin-off with strong preclinical evidence for significant breakthrough in curing prostate cancer
FREIBURG, Germany, 1-Jul-2021 — /EPR HEALTHCARE NEWS/ — Prostate cancer is the second most common cancer. According to the American Cancer Association, 1 in every 8 men will be diagnosed with it in their lifetime. In 2018 around 1.2 million men were diagnosed and there were 360,000 deaths. In Europe alone, 450,000 men are diagnosed with prostate cancer every year. But there could be a cure on the horizon.
B66, a new company in Germany and a spin-off from the University of Freiburg, Germany, has strong preclinical evidence that its research has made a significant breakthrough after 6 years of dedicated work. Professors Roland Schule, Manfred Jung and Dr Eric Metzger have pioneered research into inhibitors of the histone methyltransferase KMT9 that controls the growth of castration and enzalutamide resistant prostate tumours. These small molecular inhibitors are superior to current state-of-the-art Gold Standard drugs in preclinical models. This new treatment has the prospect of increasing survival in the normally fatal outcome of castration-resistant prostate cancer and becoming the universal therapeutic agent. In addition, the research team’s work shows that their new therapeutic agent has potentials in other cancers including colorectal, breast, bladder and the biggest of all killers, lung cancer.
Professor Roland Schule, one of the B66 founders, and a professor of Biochemistry in the University Medical Centre of Freiburg said “Our findings and approach to treatment is far in advance of anything that is going on in the world at the moment.â€
Fellow founder, Professor Manfred Jung, added “many cancer treatments cause damage to the patient’s normal cells but the B66 inhibitor does not target normal cells indicating that our treatment will have minimum side effectsâ€.
Although focused on prostate cancer right now, the B66 team’s long-term objective is to create a world-class development platform that will work on the defeat of many other types of cancer. The Freiburg region, thanks to the University, is a unique centre of leading medical knowledge. B66 will expand their work and draw in some of the skilled scientists from the area to help them work towards building a leading medical development hub.
The B66 team is already seeking commercial partners in order to forward their breakthrough work and advance as quickly as possible to defeat this major cancer.
SOURCE: EuropaWire
GenieMD to Feature Award Winning AI-based Telemedicine Platform at HIMSS19
![]() San Francisco, CA, 2019-Jan-24 — /EPR Network/ — GenieMD will be exhibiting at HIMSS19 in Orlando, Florida February 11-15, 2019. The GenieMD team will be showcasing their iVisit Telemedicine platform. GenieMD’s award-winning Telemedicine platform, driven by an AI engine, assists in the patient’s triage allowing for a streamlined consult with their physician through its HIPAA compliant text messaging, video visits and question-based asynchronous visits.
San Francisco, CA, 2019-Jan-24 — /EPR Network/ — GenieMD will be exhibiting at HIMSS19 in Orlando, Florida February 11-15, 2019. The GenieMD team will be showcasing their iVisit Telemedicine platform. GenieMD’s award-winning Telemedicine platform, driven by an AI engine, assists in the patient’s triage allowing for a streamlined consult with their physician through its HIPAA compliant text messaging, video visits and question-based asynchronous visits.
Patients are demanding convenience, expanded access and lower cost of health care. Doctors are overwhelmed with simple cases which take up most of their consult time. Clinics are struggling to manage costs in light of their patients increasingly use of alternative channels to seek treatment. To address these challenges, technology companies are innovating new ways for patients to gain access to providers. Government regulators are responding more favorably to the use of these methods including Telemedicine. Therefore, Telemedicine adoption is growing at a rapid pace.
“At GenieMD, our mission is to help improve the quality of human life by providing solutions that enable people to live healthier and longer.†– Dr. Soheil Saadat, CEO
The company offers a wide range of products and services designed to democratize healthcare globally.
Via EPR Network
More Healthcare press releases
Europe Home Diagnostics Market: Growth in Pregnancy and Ovulation Predictor Test Kits Sales Encouraging
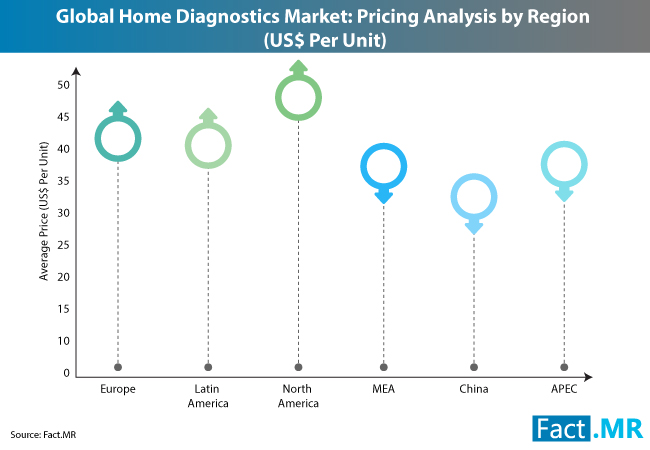
DUBLIN 2, Ireland, 18-Oct-2018 — /EPR HEALTHCARE NEWS/ — The European home diagnostics industry is realigning to foster integration with the broader customer-centric healthcare services. Growing consumer preference for home-based diagnostics, combined with innovation in online platforms, is creating sustained opportunities in this dynamic landscape. Riding on the coattails of these factors, the US$ 1.4 billion Europe home diagnostics market is likely to grow at 3.4% CAGR during the period 2018-2026.
Consumer Preference for Non-Invasive Glucose Monitoring Devices Still Remains UnmetÂ
Glucose monitoring kits and devices remain the top-selling product, accounting for over 80% revenue share of the Europe home diagnostics market. The burgeoning demand for home-based glucose monitoring devices and kits can be attributed to growing prevalence of diabetes in Europe, which is increasing across all ages according to the WHO. The Fact.MR study opines that digital devices will remain the preferred choice over cassettes among European consumers.
The broader trends pertaining to non-invasive and painless diagnostics are driving innovation in glucose monitoring device development. According to the Fact.MR study, non-invasive glucose monitoring devices are likely to witness increasing penetration in Europe, albeit at a slow pace. The report also finds that broader innovation in non-invasive devices can drive healthcare consumerism across Europe.
Request a sample of the report
https://www.factmr.com/connectus/sample?flag=S&rep_id=1892
The Fact.MR study remains optimistic on the future growth in Europe’s home diagnostics market, however, challenges prevail. “Self-testing, especially for people with type 1 diabetes, who require daily or frequent testing, remains a painful and inconvenient process. Over the decades, the innovation in glucose monitoring devices hasn’t really addressed the invasive part of the self-testingâ€, says Mr. Santosh Kumar, Senior Consultant at Fact.MR. “There is an urgent need for non-invasive glucose monitoring testing,†adds Mr. Kumar.
The Fact.MR study finds that home diagnostics manufacturers are aware of the evolving demand. Investment and research into developing minimally or non-invasive home diagnostics devices is gaining ground. “The realization that digital, and non-invasive devices are the future is dawning upon manufacturers. Market leaders, such as Siemens Healthcare and Abbott Technologies are leveraging wearable technologies and IoT to develop innovative products,†says Mr. Kumar.
Growth in Pregnancy and Ovulation Predictor Test Kits Sales EncouragingÂ
Although glucose monitoring devices and kits will account for the majority of sales, encouraging trends in other self-test kits continue to complement market growth. Pregnancy test kits and ovulation predictor test kits are likely to witness surging demand, as an increasing consumer base feels confident about self-testing. Gains are also encouraging, albeit miniscule, in other home diagnostics products, such as urinary tract infection kits and male infertility test kits.
UK Maintains its Numero Uno Position, Germany and France Remain Lucrative
The Europe home diagnostics market remains concentrated in the UK, Germany, and France. These three markets collectively account for a market share of nearly 60% revenue share of the Europe home diagnostics market. The Fact.MR study opines that these three markets will witness increasing proliferation of next-gen home diagnostics devices.
Request methodology behind the report
https://www.factmr.com/connectus/sample?flag=RM&rep_id=1892
Fact.MR study on the home diagnostics market brings interesting insights to the fore. According to the report, UK remains Europe’s largest market for home diagnostics, with revenues to the tune of US$ 280 million in 2017. The study opines that UK will maintain its numero uno position throughout the course of the forecast period 2017-2026. Germany and France will follow suit, however, sales will grow at a sluggish pace in the former.
The report also finds that home diagnostics devices that use urine and blood for testing will continue to remain pervasive in the Europe home diagnostics market. Samples devices and kits that use semen and breath are expected to witness growth during the course of the forecast period.
Buy the report
https://www.factmr.com/checkout/1892/S
SOURCE: EuropaWire
IBM Watson Market Study: IBM Watson services have been positively disrupting and reforming operations in the healthcare industry
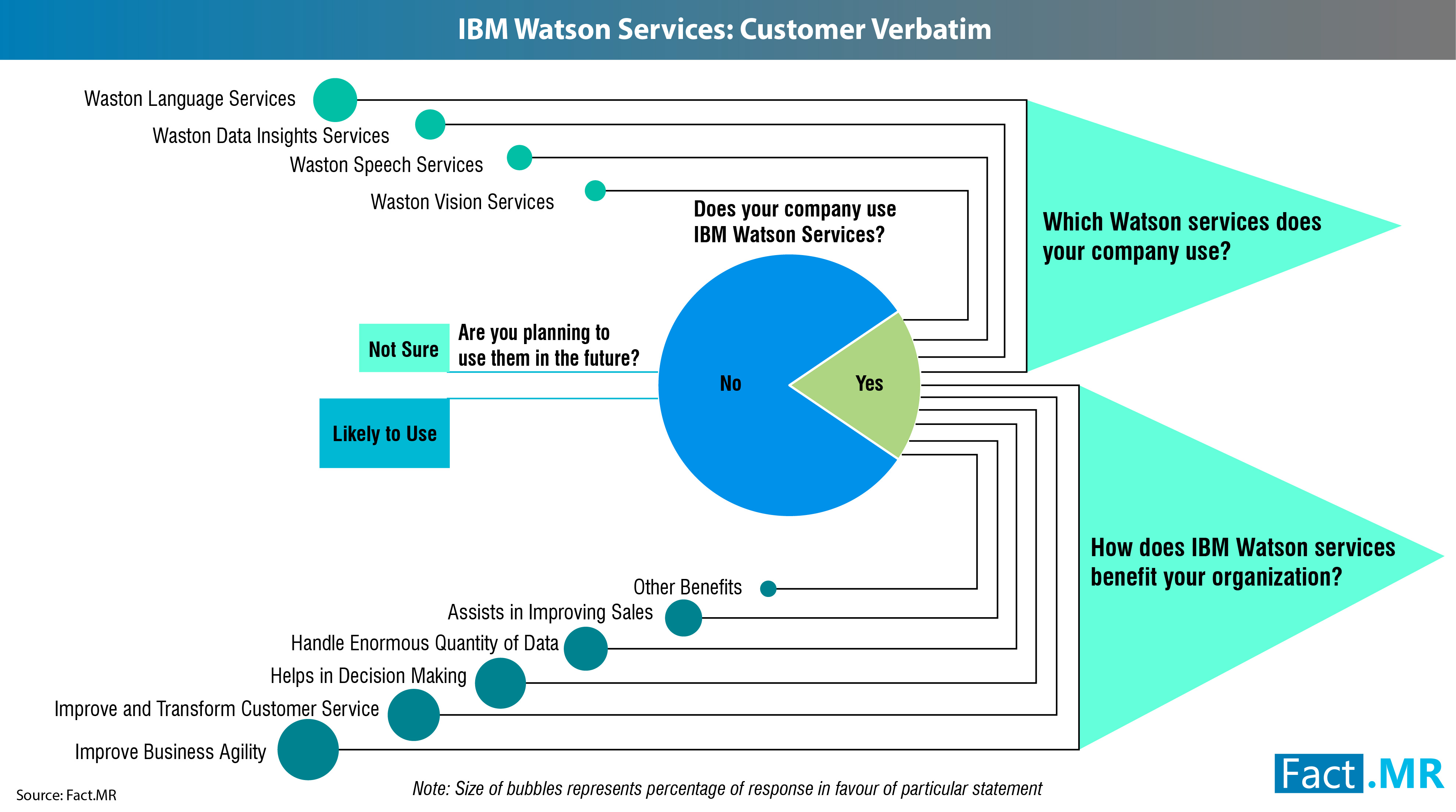
DUBLIN 2, Ireland, 10-Oct-2018 — /EPR HEALTHCARE NEWS/ — IBM Watson Services continue to get significant traction from end-users, with over 72% respondents in Europe affirmative on using IBM Watson language services, according to a Fact.MR survey. The survey is part of the comprehensive research study on IBM Watson services market which tracks the adoption of this platform across key sectors. The Fact.MR survey also reveals that use of IBM Watson vision services attracted the minimum interest, with only 48% of respondents in Europe affirmative about using it.
Fact.MR’s IBM Watson services market survey gathered data from over 500 respondents in 10 European countries. These respondents represented large-scale, SMEs, and micro organizations, with a collective IT spending budget of over US$ 50 billion. The findings of the survey were in sync with the overall expectations and use of IBM Watson services, which are highly reliant on revenue generated from language services.
Growing Realization that Cognitive Technologies Can Empower Decision-MakingÂ
The survey indicates that mitigating the gaps that exist in IT vendors’ execution from data collection to action remains the key concern of organizations. There is an increasing realization among IT vendors that cognitive technologies can empower decision-makers to make sense of colossal structured and unstructured data. The survey offers encouraging insights on the growing use of advanced analytics and cognitive computing in today’s data-driven economy, as over half of the respondents in the survey agreed to using insights- and speech-services of IBM Watson.
Request Sample of IBM Watson Services Market Report at https://www.factmr.com/connectus/sample?flag=S&rep_id=759
“Today, the requirements of organizations are multi-pronged – from gaining a holistic customer view to agile data-to-execution operations, organizations need to invest in cognitive computing and advanced technologies to stay competitive,†says Shambhu Nath Jha, Senior Consultant at Fact.MR and co-author of the study.
“IBM Watson services have been positively disrupting and reforming operations across sectors, especially healthcare. From screening patient’s structured and unstructured data to regulatory requirements and medications, IBM Watson services are witnessing extensive adoption in the healthcare sector,†added Mr. Jha. The growing adoption of IBM Watson services in healthcare is further substantiated by Fact.MR’s findings that the sector currently accounts for nearly 21% revenue share, representing a market value worth US$ 318 Mn.
Improved Business Agility and Seamless Customer Service Identified as Key AdvantagesÂ
The Fact.MR survey further reveals that improvement in business agility is the most prominent motivation driving investment in IBM Watson services. Nearly 84% of respondents currently using IBM Watson services said that there was a marked improvement in business agility since the introduction of platform in their organization. An additional 82% said IBM Watson services helped their organizations in improving and transforming customer service.
“Customer service has often remained a key concern for organizations; despite the proliferation of advanced technologies and pervasiveness of smartphones, nearly 50% of customer support calls remain unresolved,†says Shambu Nath Jha. “The integration of artificial intelligence (AI) can mitigate this challenge, and offer seamless support for Tier 1 queries. The system can also effectively gauge the queries that require human intervention, and escalate those queries to a customer support executive if necessary,†adds Mr. Jha.
Know the methodology behind the reportÂ
https://www.factmr.com/connectus/sample?flag=RM&rep_id=759
Effective decision-making, handling colossal structured and unstructured data, and sales assistance are among the other key advantages of using IBM Watson services, according to the Fact.MR survey. “Effective decision-making entails a deep understanding of the unique needs and requirements of customers. Today, businesses are hard-pressed to understand the unique characteristics of their customers, and effectively map their existing and upcoming products to meet the overt and covert needs. IBM Watson can provide sophisticated recommendations that can facilitate effective decision-making,†Mr. Jha added.
Fact.MR’s report on the global IBM Watson services market projects a surge in investments to mitigate cybersecurity risks. According to Fact.MR, a majority of organizations have some level of cybersecurity apparatus in place. However, as Mr. Jha concludes, “tackling the potent cybersecurity threats will require a comprehensive ecosystem, rather than a symbolic front-line defense.â€
The IBM Watson Services Market Report is Available for Direct Purchase at
https://www.factmr.com/checkout/759/S
SOURCE: EuropaWire
Alliance Life Sciences to present expanded portfolio of pricing & market access services at the World Pharma Pricing and Market Access Congress 2018

SOMERSET, N.J. / LONDON, UK, 19-Mar-2018 — /EuropaWire/ — Alliance Life Sciences, a leading global life sciences consulting, pricing data and technology provider, announced that it is a sponsor at the World Pharma Pricing and Market Access Congress 2018 taking place March 20-21 in London, United Kingdom. As part of the largest pharmaceutical commercialization organization that services more than 300 of the industry’s top pharmaceutical, biotech and medical device companies, Alliance will present its expanded portfolio of pricing and market access services. The focus will be on pricing and reimbursement data managed by PricentricOne and pricing governance, reference and tender pricing software manage by PriceRight.
Now in its 12th year the World Pharma Pricing and Market Access Congress brings together the most innovative leaders and organizations to discuss all aspects of evidence, access and pricing. This year’s event will feature over 150 speakers and a greater range of topics being covered including Big Data & AI, rare diseases, advanced therapies and other emerging, innovative areas.
Alan Crowther, President of Global Markets noted “Alliance Life Sciences is pleased to be a sponsor of the World Pharma Pricing & Market Access congress, particularly this year. We have made a number of investments in our people and infrastructure in Europe. Our acquisition by Water Street partners in 2017 allowed us to offer enhancements in our Market Access offerings too. Europe continues to be our focus market and World Pharma Pricing & Market Access congress is a great venue to showcase our expended capabilitiesâ€.
SOURCE: EuropaWire

Prof. Lucien Abenhaim: There is an incredible challenge for the sustainability of health systems across the world

LONDON, Dec-3-2017 — /EuropaWire/ — On December 5-6, 2017, 200 high-level experts from government, academia, biopharmaceutical developers and patient organisations convene in London (UK) to address the mounting challenge of accessibility and patient access to high-value, high-cost medical therapies.
The not-for-profit executive forum is organised by the London School of Hygiene and Tropical Medicine and analytica|LASER, a global research consultancy, to investigate topical questions around drug policy, global health systems sustainability and innovation funding through a combination of case studies, panel sessions, and roundtables. Delegations are registered from over 70 public and private organisations, including the World Health Organization, as well as health systems representatives from Belgium, England, Estonia, Croatia, France, Germany, Italy, Norway, Sweden, the United States and Wales. Multiple patient organisations support the meeting to ensure that the patient voice is reflected in all deliberations.
“Access to life-saving drugs is not a new challenge. The paradigm of accessibility was changed completely in the face of the deadly HIV epidemic. This time, however, accessibility concerns almost every domain of medicine such as cancer, auto-immune diseases and rare diseases. We must act now,†says Prof Peter Piot, Director of the London School of Hygiene & Tropical Medicine and former head of UNAIDS.
“There is an incredible challenge for the sustainability of health systems across the worldâ€, says Prof. Lucien Abenhaim, London School of Hygiene & Tropical Medicine and Chairman of Analytica Laser. “It is critical that drug developers and health system engage on actionable proposals that address concerns around value, cost and overall access to innovation for the patients who need it. We are at an inflection point – the paradigms of the 1990’s can no longer guide us,†he adds.
The main plenary on December 6th features scheduled remarks by over a dozen authorities in the field, including Professor Baron Peter Piot (Director, London School of Hygiene & Tropical Medicine and former Head, UNAIDS), Dr. Olivier Brandicourt (Chief Executive Officer, Sanofi), Dr. Sarah Garner (World Health Organization), Prof. Dominique Le Guludec (President, French HAS), Sir Andrew Dillon (Chief Executive, National Institute for Health and Care Excellence UK) and Dr. Harold Paz (Chief Medical Officer, Aetna). The plenary is preceded by a scientific seminar on December 5th which gathers globally renowned academics to explore new frontiers on the methodology for the appraisal and valuation of high-value medicines.
Please contact the organisers for more information on the programming and media access, or refer to www.accessibility-symposium.org
SOURCE: EuropaWire

MEDLAB Series Europe, 13-15 Sep 2017, will cover Point of Care Testing (POCT), Histopathology, Lab Management, Microbiology and Haematology
Spain, 04-May-2017 — /EuropaWire/ — After many years of operating successful MEDLAB events around Africa, Asia and the Middle East, Informa Life Sciences Exhibitions, the worlds leading publishing and exhibitions company, has announced that the MEDLAB Series will be expanding its presence into Europe. Taking place at the Barcelona Congress Centre in Barcelona, Spain, from 13-15 September 2017, more than 3,000 industry professionals are expected to attend Europe’s leading event for laboratory management and diagnosis.
With the European In-Vitro Diagnostics (IVD) market expected to reach USD 15.5 billion by 2024, a platform such as MEDLAB presents a huge opportunity for global laboratory industry leaders, including manufacturers, dealers and distributors, to make inroads into the European market. Housing international exhibitors and covering 2,000 sqm of exhibition space, MEDLAB Europe will give visitors from across the world an opportunity to access cutting-edge laboratory products, next-generation technology, innovative services and world-class educational content.
According to Tom Coleman, Group Exhibition Director, MEDLAB Series: “The launch of MEDLAB Europe is in line with our global expansion strategy for our MEDLAB series of events. The increasing prevalence of chronic diseases, rising geriatric population coupled with the rising awareness towards early diagnosis, has positioned the European IVD and medical laboratory market as a critical market for manufacturers, services providers, and dealers and distributors from across the globe. MEDLAB Europe will generate substantial value for our customers and partners by driving further product innovation and deeper engagement in these specific markets.â€
Over the four-day event, MEDLAB Europe will also offer a multi-disciplinary congress tackling current challenges and developments key to the European market, and leveraging the true potential of laboratory testing to dramatically improve patient outcomes across the continent.
The conference programme covers five main tracks including Point of Care Testing (POCT), Histopathology, Lab Management, Microbiology and Haematology. From new methods of effective lab management to the development of techniques in detecting diseases, the conferences will also review the expanding role of the laboratory medicine and discuss partnership between a clinician and a lab professional in providing delivery of care to every patient.
“The scientific programme at MEDLAB has been carefully designed in collaboration with some of the brightest minds in the medical laboratory industry in order to have a real impact on improving the health and wellbeing of patients across the region,†said Coleman.
For more information about the MEDLAB Europe Exhibition & Congress, please visit: www.medlabeurope.com.
SOURCE: EuropaWire
Multispectral Optoacoustic Tomography (MSOT) can now more reliably detect early stages of inflammation
GERMANY, Mar-30-2017 — /EPR HEALTHCARE NEWS/ — Ulcerative colitis and Crohn’s disease are the most common inflammatory bowel diseases (IBD). Globally, more than 4 million people suffer from recurring inflammation of the intestinal mucosa. To date, non-invasive methods have not been able to detect these forms of IBD reliably. Researchers at the University Hospital Erlangen (Germany) have now shown for the first time how Multispectral Optoacoustic Tomography (MSOT) can more reliably detect early stages of inflammation when compared with other non-invasive diagnostic methods. These results have recently been published in the New England Journal of Medicine.
Evaluating the inflammatory activity of the intestines early and accurately helps to prevent complications associated with IBD and guide an optimal therapy regime. Currently, doctors frequently rely on an invasive endoscopy procedure to assess the intestinal mucosa of patients with IBD. Additionally, non-invasive imaging methods are increasingly used in the clinic. For example, ultrasound allows clinicians to examine the structure, thickness and blood perfusion of the intestinal wall with changes in perfusion patterns indicating an onset of inflammation.
“With the MSOT procedure, we additionally use laser light, by which we can detect inflammation earlier than previously possible with ultrasoundâ€, explains Prof. Dr. Maximilian Waldner, Professor at the University Hospital Erlangen. “The earlier we detect signs of an upcoming flare – i.e., a change in blood content – the quicker we can react and start a preventive treatmentâ€. The MSOT technology was developed by iThera Medical in Munich, and was first used on IBD patients last year.
The researchers of the University Hospital Erlangen have recently published their results in the renowned New England Journal of Medicine. “We examined 108 patients with Crohn’s disease using the MSOT technology and compared the results to established non-invasive diagnostic procedures as well as endoscopy and histologyâ€, says Prof. Waldner. “We were able to demonstrate that hemoglobin values in the tissue measured with MSOT indicate very accurately the inflammatory activity in the intestines. The method appears to be superior to other non-invasive procedures, as MSOT for the first time was able to detect even low levels of inflammation without having to perform endoscopyâ€.
Dr. Ferdinand Knieling, a physician at the University Hospital Erlangen, adds: “We are hoping that this principle can be applied to many applications and, in particular, also be used to enable safe evaluations of children and adolescents. With this new examination method, many of the currently invasive procedures might become obsolete.â€
In optoacoustic imaging, the physician scans the intestines transabdominally through the skin, applying pulsed laser light in the near-infrared region. The energy of the light absorbed in tissue is converted into acoustic signals which are then acquired by an ultrasound detector. Christian Wiest, CEO of iThera Medical, comments: “MSOT is a new diagnostic imaging modality that can detect changes in tissue composition associated with a variety of diseases, without the use of contrast agents. The technology has already been applied in other clinical feasibility studies, e.g. for the detection of melanoma metastasis in sentinel lymph nodes or for the assessment of suspicious breast lesionsâ€.
The results of the IBD study at the University Hospital Erlangen were published on March 30th 2017 in the New England Journal of Medicine (Volume 376, Issue 13, pg.1292-1294).
SOURCE: EuropaWire
Global leader in serialization solutions Systech to attend the annual pharmaceutical congress CPhI Worldwide in Barcelona, 4 – 6 Oct 2016
 BRUSSELS, 28-Sep-2016 — /EuropaWire/ — Systech International, a global leader in brand protection technologies, will partner with Sharp Packaging and SINEL SYSTEMS, part of the Autajon Group, at CPhI Worldwide, the annual pharmaceutical congress held in Barcelona from 4 – 6 October 2016.
BRUSSELS, 28-Sep-2016 — /EuropaWire/ — Systech International, a global leader in brand protection technologies, will partner with Sharp Packaging and SINEL SYSTEMS, part of the Autajon Group, at CPhI Worldwide, the annual pharmaceutical congress held in Barcelona from 4 – 6 October 2016.
Systech will be part of Sharp’s panel discussion addressing delegates on the theme of “Serialization: Optimizing Your Supply Chain Means Optimizing Your Partners.†The presentation will explore the critical path to supply chain readiness, risk assessment, benchmarking and serialization risk mitigation options. The briefing will take place in the Pharma Insight Briefing room on Tuesday, 4 October 2016 at 11:30am CEST.
Systech will also showcase its UniSeries™ serialization software offered as an integrated solution within Autajon’s exhibit booth #2P16 (Hall 2).
CPhI Worldwide, together with co-located events ICSE, InnoPack, P-MEC and FDF, will host more than 36,000 visiting pharma professionals from 4-6 October at the Fira Gran Via, Barcelona, Spain. More than 2,500 exhibitors from 150+ countries gather at the event to network and take advantage of more than 100 free industry seminars. Every sector of the pharmaceutical market is represented under one roof, this year in a new location in Barcelona.
With thousands of implementations across the globe, Systech is the world’s most trusted brand protection expert, and its UniSeries offering is the industry standard for serializing products. UniSeries is offered as an integrated solution with both Sharp Packaging and Autajon Group’s hardware. Systech’s patented, configurable software offers manufacturers the flexibility to adapt to new regulations without extensive reworking or revalidation, thereby reducing implementation, training and support costs.
“We are recognized as the world leader in serialization and brand protection,†states Manfred Voglmaier, Senior Director Sales Europe, Systech International. “With more than 30 years of experience in providing highly configurable solutions for serialization across a variety of industries ― along with our partners Sharp Packaging and Sinel ― gives us a unique insight for this panel.â€
Systech’s products are in greater demand than ever before since it is estimated that over 15,000 production lines for prescription medication in Europe must be fitted with a serialization system over the next three years. The European Commission adopted a regulation on October 2, 2015, under which the serialization of prescription medicine packages will become mandatory in all EU countries by 2019.
Show exhibit hours are on Tuesday, 4 October – Thursday, 6 October. Systech representatives will be available in the Sharp Packaging booth #3L56 (Hall 3) during the event, as well as in the Autajon booth #2P16 (Hall 2). They will showcase Systech’s software solutions on sample packaging lines and via live demos throughout the event.
SOURCE: EuropaWire


